Copper corrosion inhibitors play an essential role in maintaining the reliability and efficiency of modern water systems. In industrial environments where copper and copper alloys are widely used, uncontrolled corrosion can lead to equipment degradation, reduced heat transfer efficiency, and costly downtime. From cooling towers to closed-loop systems and process applications, managing copper corrosion is a fundamental part of a well-designed water treatment program.
At its core, copper corrosion is a chemical and electrochemical process that occurs when the copper surface reacts with surrounding liquids, dissolved oxygen, and other corrosive species. Over time, this interaction can produce corrosion products, release copper ions into the system, and negatively affect other materials such as steel through deposition and galvanic interactions. These effects are not always immediately visible, but they can significantly impact long-term system performance.
The use of copper corrosion inhibitors helps control these reactions by forming a protective barrier on the metal surface and stabilizing system chemistry. When properly selected and applied, a corrosion inhibitor can reduce corrosion rates, improve resistance to aggressive conditions, and extend the service life of critical components. However, effectiveness depends on multiple factors including pH, chemical conditions, contaminant presence, and overall system design.
This article explores how copper corrosion occurs, how inhibitors function, and how they are applied and monitored in industrial water systems. Understanding these principles is essential for selecting the most suitable solution and maintaining consistent protection across a wide range of operating conditions.
Understanding Copper Corrosion in Water Systems
Copper corrosion in water systems is an electrochemical process influenced by the interaction between the metal surface, surrounding solution, and various corrosive species. While copper is known for its natural resistance, it is not immune to degradation, especially when exposed to changing chemical conditions and aggressive environments. Understanding how corrosion develops is essential to controlling its occurrence and minimizing long-term damage.
Electrochemical Nature of Copper Corrosion
At a fundamental level, copper corrosion occurs through oxidation and reduction reactions at the surface of the metal. When copper is exposed to water containing dissolved oxygen, it can oxidize to form copper ions, which then enter the solution. This process represents the anodic reaction, while a corresponding reduction reaction, often involving oxygen, occurs at another location on the surface.
These reactions may seem minor at first, but over time they can lead to measurable corrosion rates, especially in systems where water chemistry is not stable. Even in relatively clean environments such as distilled water, corrosion can still occur if oxygen or other reactive species are present.
Role of Oxygen and Corrosive Species
Dissolved oxygen is one of the primary drivers of copper corrosion, but it is not the only factor. Other corrosive species, including ammonia, can significantly accelerate the process. Ammonia, for example, can complex with copper ions, increasing their solubility and promoting further metal loss.
Additionally, the presence of dissolved salts, varying pH, and other metal ions can create localized conditions that enhance corrosion activity. These interactions are often system-specific, making it important to evaluate water chemistry as a whole rather than focusing on a single parameter.
Formation of Corrosion Products
As copper corrodes, it forms a corrosion product, typically composed of copper oxide layers on the surface. In some cases, these oxide films can provide partial protection by limiting further exposure of the underlying metal. However, this protection is not always stable.
Under certain conditions, such as fluctuating pH or high concentrations of aggressive species, the oxide layer can break down or become porous. This allows corrosion to continue and may result in the release of additional copper ions into the system. These ions can travel through the system and deposit on other materials, including steel, leading to secondary corrosion concerns.
Because corrosion behavior is highly dependent on environmental conditions, it must be carefully evaluated within the context of each system. Changes in water chemistry, operating conditions, or contaminant levels can significantly alter the extent of corrosion, making ongoing monitoring and control essential.
Key Factors That Influence Copper Corrosion
Copper corrosion in water systems does not occur under a single fixed condition. Instead, it is influenced by a range of variables related to water chemistry, system design, and operational practices. Understanding these factors helps determine the extent of corrosion and supports the selection of appropriate treatment strategies.
Below are the primary factors that influence how and when copper corrosion may occur:
- Water Chemistry and Concentration Levels
The concentration of dissolved substances in a solution, including salts, gases, and treatment chemicals, directly impacts corrosion behavior. Variations in pH, alkalinity, and ionic strength can shift the balance between protective film formation and metal dissolution. In many systems, even small changes in concentration can significantly affect corrosion rates. - Hardness and Dissolved Solids
Water hardness, primarily from calcium and magnesium, can influence scaling and corrosion simultaneously. In some cases, hardness can contribute to the formation of protective deposits, while in others it may interfere with inhibitor performance. The presence of dissolved solids in liquids can also alter how corrosion processes develop on the metal surface. - Presence of Contaminants and Reactive Species
The presence of contaminants such as ammonia, chlorides, or other reactive compounds can accelerate corrosion. These substances may interact with copper ions, destabilize protective films, or create localized aggressive conditions. When multiple contaminants are present, their combined effect can be more severe than any single component alone. - System Materials and Mixed Metallurgy
Many industrial water systems contain a variety of materials, including copper, steel, and other alloys. When different metals are in direct contact or connected through the same water circuit, galvanic interactions can develop. This can increase the likelihood that copper will corrode or that dissolved copper will deposit onto other surfaces. - Exposure and Contact Conditions
The degree to which copper components are exposed to flowing water, air, or chemical additives affects corrosion behavior. Areas with high turbulence, stagnant zones, or repeated cycles of wet and dry conditions can experience different corrosion patterns. The duration and type of contact between the metal and the surrounding environment also play a role. - System Operation and Frequency of Changes
Operational factors such as temperature fluctuations, flow rates, and maintenance practices influence corrosion. Systems that are adjusted frequently, or that experience inconsistent operating conditions, may see more variable corrosion behavior. Over time, these changes can increase the overall extent of damage.
Because these factors often act together, copper corrosion must be evaluated as part of a complete system analysis. A change in one variable can influence several others, making it important to consider the full operating environment when developing a corrosion control program.
How Copper Corrosion Inhibitors Work
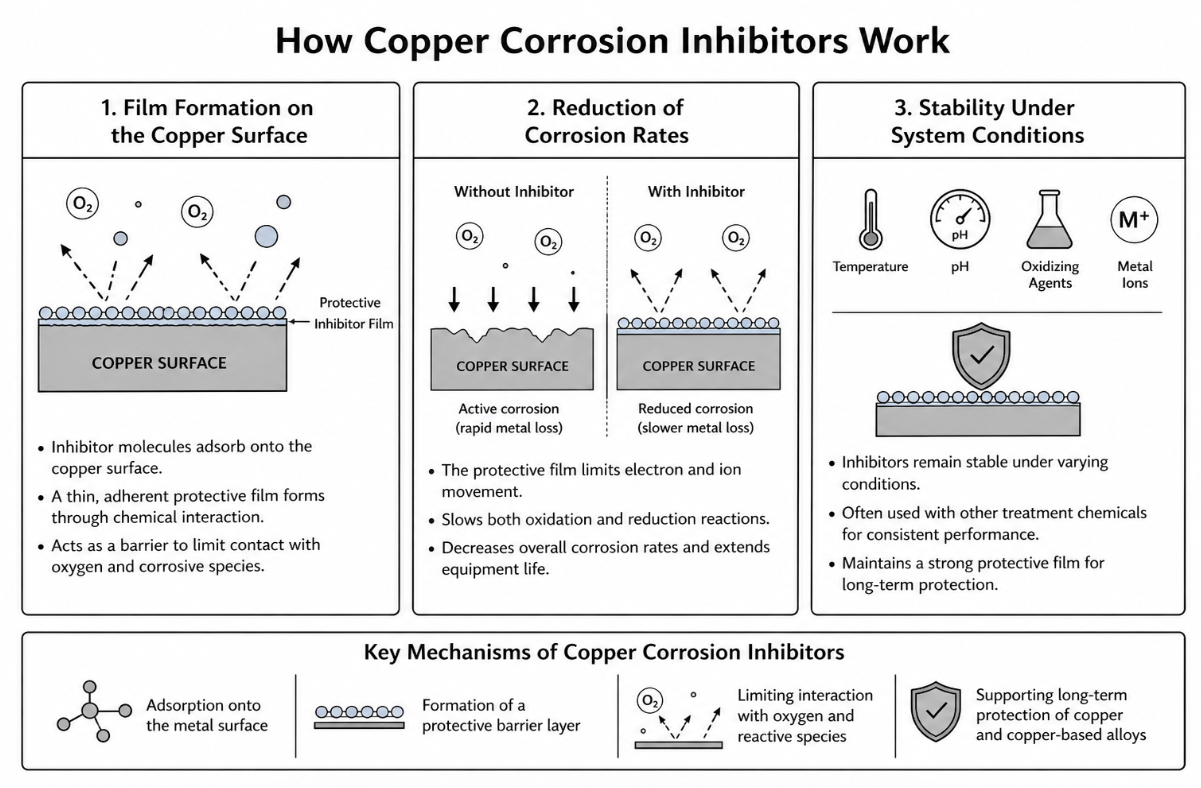
Copper corrosion inhibitors are designed to control the electrochemical reactions that lead to metal loss by forming a barrier between the copper surface and the surrounding environment. Rather than eliminating corrosion entirely, these treatments reduce the rate at which reactions occur, helping maintain system integrity under a wide range of operating conditions.
Film Formation on the Copper Surface
The primary mechanism of most copper corrosion inhibitors is the formation of a thin, adherent protective coating on the metal surface. This coating develops through chemical interaction between the inhibitor molecules and the copper, creating a stable layer that limits exposure to dissolved oxygen and other corrosive species.
This process can be described as a type of formation reaction, where the inhibitor adsorbs onto the surface and bonds with the metal. Once established, this layer acts as a physical and chemical barrier, reducing the ability of reactive substances to reach the copper beneath.
Reduction of Corrosion Rates
By limiting direct interaction between the metal and its environment, a corrosion inhibitor helps decrease overall corrosion rates. The protective film reduces the movement of electrons and ions that drive corrosion reactions, effectively slowing both oxidation and reduction processes.
This increased resistance to corrosion is especially important in systems where copper is continuously exposed to flowing water, fluctuating temperatures, or changing chemical conditions. When properly maintained, inhibitor programs can keep corrosion activity at controlled, predictable levels.
Stability Under System Conditions
The effectiveness of copper corrosion inhibitors depends on their ability to remain stable under real-world operating conditions. Factors such as pH, temperature, oxidizing agents, and the presence of other metal ions can influence how well the protective layer performs.
In many cases, inhibitors are used in combination with other treatment chemicals, such as dispersants or scale inhibitors, to maintain consistent performance. This integrated approach helps ensure that the protective film remains intact even as system conditions change.
Common mechanisms associated with inhibitor performance include:
- Adsorption onto the metal surface
- Formation of a protective barrier layer
- Limiting interaction with oxygen and reactive species
- Supporting long-term protection of copper and copper-based alloys
Because each system is unique, selecting the right copper corrosion inhibitors requires careful consideration of water chemistry, materials of construction, and operational demands. When properly applied, these treatments provide a reliable method to protect copper components and extend the service life of critical equipment.
Types of Copper Corrosion Inhibitors and Chemistries
Copper corrosion inhibitors are available in several chemical forms, each designed to address specific system conditions and treatment goals. Selecting a suitable inhibitor depends on factors such as water chemistry, system design, and compatibility with other treatment chemicals. While many formulations share similar objectives, their performance can vary based on how they interact with the system environment.
Common Inhibitor Chemistries
The most widely used copper inhibitors are organic compounds that form protective films on the metal surface. Among these, triazole-based chemistries are commonly applied due to their effectiveness and stability.
| Inhibitor Type | Common Use | Key Advantage | Consideration |
|---|---|---|---|
| Benzotriazole (BZT) | Cooling water, closed loops | Strong film formation on copper surfaces | Can be affected by strong oxidizers |
| Tolyltriazole (TTA) | Cooling towers, mixed-metal systems | Improved stability in certain environments | Requires proper dosing control |
| Phosphate-based blends | Multi-metal systems | Helps protect both copper and steel | Performance depends on pH and scaling potential |
| Blended formulations | Complex industrial systems | Balanced protection across multiple materials | Must be matched to system chemistry |
These inhibitors work by interacting with the copper surface to create a stable barrier that limits corrosion. In many cases, they are used in combination with other treatment chemicals to enhance overall system performance.
Role of Acids and Laboratory Reagents
In laboratory settings, copper corrosion behavior is often studied using controlled environments that include acid solutions such as hydrochloric acid. These conditions help researchers evaluate how quickly copper may corrode and how effective different inhibitors are under aggressive conditions.
Compatibility and System Considerations
Choosing the right inhibitor involves more than selecting a single chemistry. The inhibitor must be compatible with:
- Existing treatment chemicals
- System metallurgy, including copper and other alloys
- Operating conditions such as temperature and flow
- The presence of oxidizing or reducing agents
Additionally, inhibitor performance may be influenced by redox conditions, where reduction and oxidation reactions occur simultaneously. These interactions can impact how well the protective film forms and how long it remains effective.
Because no single inhibitor is ideal for every application, treatment programs are typically designed based on system-specific requirements. Proper selection and application help ensure consistent protection and minimize the risk of unintended interactions within the system.
Application in Industrial Water Systems
Copper corrosion inhibitors are applied across a wide range of industrial systems, where copper and copper-based alloys are used for heat transfer, piping, and process equipment. Their role is not isolated, they are part of a broader treatment process that must be properly designed, implemented, and evaluated to ensure consistent performance.
Cooling Towers
In cooling towers, copper corrosion control is especially important due to continuous exposure to oxygen, heat, and varying water chemistry. These towers often contain mixed metallurgy, including copper, steel, and other materials, which increases the risk of galvanic interactions.
Inhibitors are typically dosed into the recirculating water to maintain a target concentration that supports film formation on the copper surface. Performance is often evaluated through routine testing, with results indicating whether adjustments are needed to maintain protection under changing operating conditions.
Closed Loop Systems
Closed loop systems generally operate under more stable conditions, but corrosion can still occur if oxygen ingress, chemical imbalance, or contamination is present. In these systems, copper corrosion inhibitors help maintain a stable protective layer over long periods.
Because water is often stored and recirculated with minimal replacement, maintaining consistent inhibitor levels is critical. Even small deviations in chemistry can influence corrosion behavior, making monitoring an essential part of the treatment strategy.
Boiler and Condensate Systems
In boiler and condensate return systems, copper components are often found in feedwater lines, heat exchangers, and return piping. Corrosion in boilers can lead to the release of metal ions, which may deposit elsewhere in the system.
Inhibitors are used to facilitate protection in these environments by stabilizing copper surfaces and reducing the likelihood of metal transport. Proper application ensures that corrosion is controlled without interfering with other aspects of boiler water chemistry.
System Integration and Performance Considerations
Across all applications, successful use of copper corrosion inhibitors depends on proper integration into the overall treatment program. This includes:
- Maintaining appropriate inhibitor concentration levels
- Ensuring compatibility with other chemicals in the system
- Monitoring performance through regular testing and analysis
- Adjusting treatment based on system conditions and results
Because industrial water systems vary widely across different industries, inhibitor programs must be tailored to specific operating environments. A well-managed program supports consistent protection, reduces maintenance concerns, and helps extend the life of critical equipment.
Monitoring and Performance Evaluation
The effectiveness of a copper corrosion control program depends on consistent monitoring and accurate performance evaluation. Even well-selected inhibitors require ongoing verification to ensure that protection remains stable under changing system conditions. Without routine testing, corrosion issues may develop gradually and only become evident after significant damage has occurred.
A key aspect of monitoring is measuring inhibitor residual levels in the system. These values are typically measured using field test kits or laboratory analysis to confirm that the target concentration is being maintained. When residual levels are decreased, it may indicate higher demand, system losses, or changes in water chemistry that require adjustment.
Common monitoring approaches include:
- Residual inhibitor testing
Regular testing of water samples helps determine whether sufficient inhibitor is present to maintain protection. Consistent readings are important for ensuring stable system performance. - Corrosion rate evaluation
Corrosion coupons or probes are often used to measure actual metal loss over time. These methods help quantify corrosion activity and provide a direct indication of system conditions. - Water chemistry analysis
Parameters such as pH, dissolved oxygen, and contaminant levels are routinely evaluated. These values influence inhibitor performance and help identify conditions that may increase corrosion risk. - Visual inspection and system checks
Inspection of pipe surfaces and equipment can reveal signs of corrosion, deposition, or fouling. These observations often support analytical data and provide additional insight into system behavior.
Data collected from these methods should be reviewed on a consistent basis to identify trends and support informed decision-making. When results indicate changes in corrosion activity, adjustments to the treatment program can be made to facilitate improved performance.
Overall, monitoring is not a one-time activity but an ongoing process that helps maintain control over corrosion. By combining analytical testing with practical system observations, operators can better understand system behavior and ensure that copper protection remains effective over time.
ETI’s Role in Supporting Copper Corrosion Control Programs
Managing copper corrosion in industrial water systems requires more than selecting a single product. It involves aligning the right chemistry, system conditions, and operational practices to maintain long-term protection. For water treatment professionals, having a reliable partner behind these programs can make a significant difference in performance and consistency.
Eastern Technologies, Inc. supports water treatment companies, distributors, and OEMs by providing both chemical solutions and technical backing, without competing for end-user business. This partner-first model allows professionals to deliver effective copper corrosion control programs with confidence, backed by a manufacturer that understands real-world system challenges.
ETI’s capabilities directly related to copper corrosion control include:
- Custom-formulated corrosion inhibitor programs
ETI develops tailored copper corrosion inhibitors and blended formulations designed for specific system chemistry, metallurgy, and operating conditions. This ensures each program is suitable for the application rather than relying on generic, off-the-shelf chemistry. - Integrated cooling and boiler treatment solutions
Copper corrosion control is delivered as part of a complete treatment program that includes dispersants, scale control, and oxygen management. This integrated approach helps protect both copper and steel components across cooling towers, boilers, and closed loop systems. - Compatibility with biocide programs
With a broad portfolio of oxidizing and non-oxidizing biocides, ETI helps ensure inhibitor performance remains stable in systems where microbial control is also required. This is critical in environments where chemical interactions can impact protective film formation. - Technical support and system evaluation
ETI provides guidance to help evaluate water chemistry, operating conditions, and corrosion risks. This includes support in selecting the right inhibitor combination, optimizing dosing strategies, and adjusting programs based on system performance. - Laboratory and field support tools
Analytical support such as water analysis, corrosion monitoring, and deposit evaluation helps determine how well a treatment program is performing and where improvements can be made. - Manufacturing reliability and flexibility
ISO 9001-certified production, flexible packaging, and consistent supply help ensure that treatment programs can be implemented reliably across different locations and system types.
Because industrial water systems vary widely across industries, ETI works as an extension of its partners’ technical capabilities. This support helps ensure that copper corrosion control programs remain effective even as system conditions change.
If you are looking to strengthen your copper corrosion control programs, contact ETI Water to explore customized solutions and technical support designed to help you succeed.
Frequently Asked Questions (FAQs)
Can copper corrosion occur in distilled water?
Yes, copper corrosion can still occur in distilled water if dissolved oxygen or other reactive species are present. Even without high mineral content, corrosion may develop under certain chemical conditions.
Can household substances like vinegar dissolve copper corrosion?
Yes, mild acid solutions such as vinegar can dissolve certain copper corrosion products, especially surface oxides. However, this is not a controlled or suitable method for industrial systems.
How are copper corrosion inhibitors typically evaluated?
Performance is generally evaluated through water samples, corrosion rate measurements, and residual inhibitor testing. These methods help determine the effectiveness of the treatment under actual system conditions.
Why is copper corrosion a concern in mixed-metal systems?
Copper corrosion can release metal ions that deposit onto other materials like steel, creating additional corrosion risks. This occurrence is especially evident in systems where multiple metals are in contact.
How should treated systems be maintained during downtime?
Systems that are stored or taken offline should be properly protected to prevent corrosion during inactivity. This is commonly achieved by utilizing specialized inhibitors or layup programs designed for idle conditions.





