Maintaining proper water chemistry is essential for protecting industrial equipment, supporting treatment efficiency, and maintaining a stable pH level across water systems. One of the most important parameters in industrial water management is alkalinity, which reflects water’s ability to neutralize acids and resist sudden changes in pH. Because of its direct impact on corrosion control, scaling prevention, and treatment stability, alkalinity testing plays a critical role in both industrial water treatment and wastewater treatment operations.
Accurate alkalinity testing helps water treatment professionals evaluate buffering capacity, monitor changing water conditions, and make informed treatment adjustments before operational problems develop. Whether managing cooling towers, boiler systems, or biological wastewater treatment processes, understanding alkalinity levels can improve system performance, reduce mineral build up, and support long-term reliability in industrial equipment.
Understanding Alkalinity in Water Systems
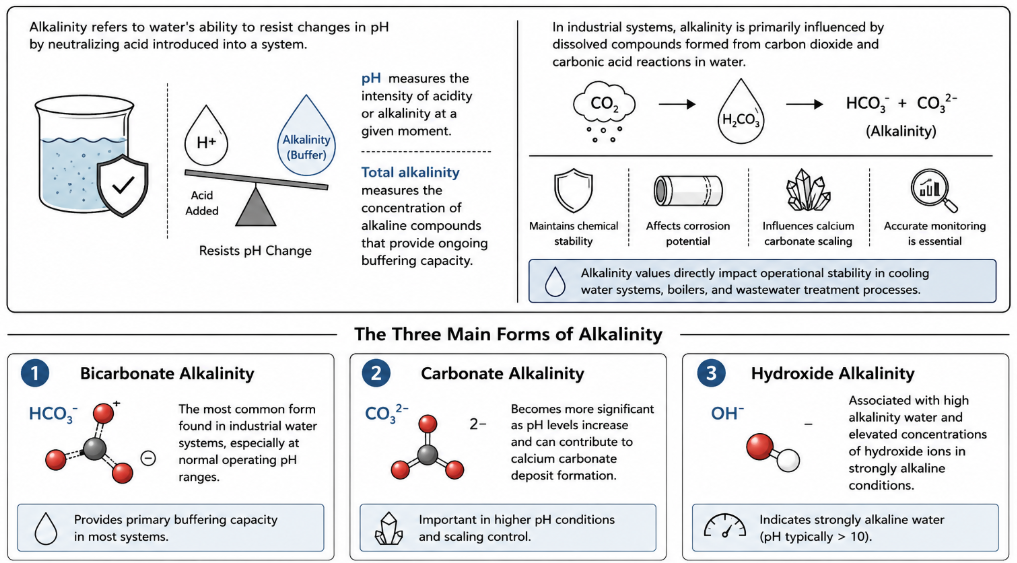
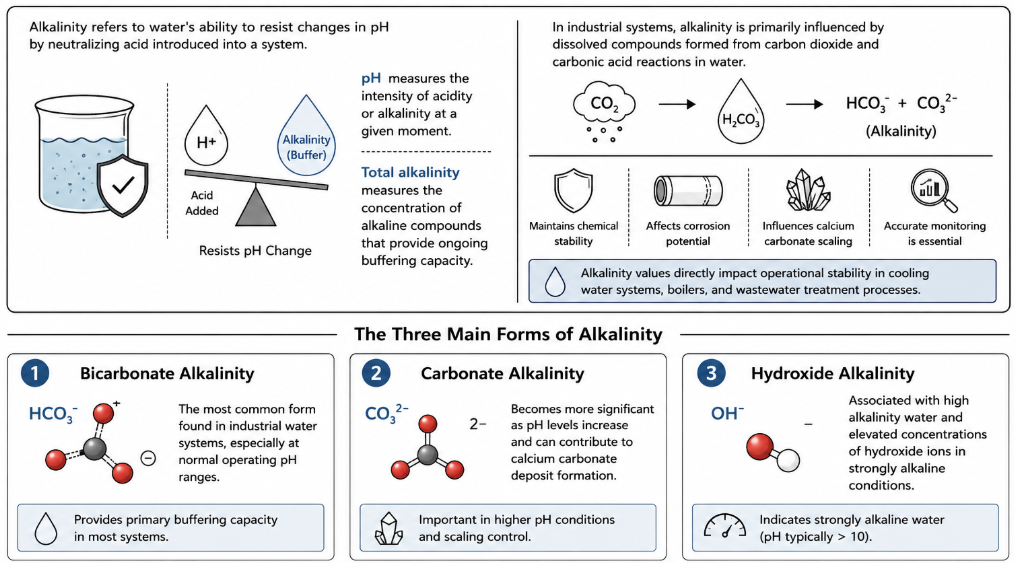
Alkalinity refers to water’s ability to resist changes in pH by neutralizing acid introduced into a system. In industrial water treatment, alkalinity acts as a buffer that helps maintain chemical stability and protects equipment from sudden pH fluctuations. While pH measures the intensity of acidity or alkalinity at a given moment, total alkalinity measures the concentration of alkaline compounds that provide ongoing buffering capacity within a water sample.
In most industrial systems, alkalinity is primarily influenced by dissolved compounds formed from carbon dioxide and carbonic acid reactions in water. These compounds determine the sample’s alkalinity and affect treatment performance, corrosion potential, and calcium carbonate scaling tendencies. Because alkalinity values directly impact operational stability, accurate monitoring is essential in cooling water systems, boilers, and wastewater treatment processes.
The Three Main Forms of Alkalinity
The three main forms of alkalinity commonly measured in water treatment systems include:
- Bicarbonate alkalinity, the most common form found in industrial water systems, especially at normal operating ph ranges.
- Carbonate alkalinity, which becomes more significant as ph levels increase and can contribute to calcium carbonate deposit formation.
- Hydroxide alkalinity, associated with high alkalinity water and elevated concentrations of hydroxide ions in strongly alkaline conditions.
The proportion of bicarbonate, carbonate, and hydroxide compounds present in a water sample influences how effectively the system can maintain stability and resist acidic conditions. Understanding these main forms helps water treatment professionals make informed decisions when determining treatment adjustments and operational control strategies.
Common Alkalinity Testing Methods
Several alkalinity testing methods are used throughout industrial and laboratory water treatment applications. The selected method often depends on the required accuracy, operating environment, and the type of system being monitored. In many facilities, alkalinity measurement is performed routinely to support corrosion prevention, scaling control, and process efficiency.
Titration Testing
Titration remains one of the most widely used methods for alkalinity testing in industrial water systems. This method involves adding a controlled acid solution to a water sample until a specific endpoint is reached. During the procedure, indicators and reagents are used to identify a color change that signals the completion of the reaction.
Phenolphthalein alkalinity, commonly called p alkalinity, is measured first to determine the presence of hydroxide and carbonate compounds. Total alkalinity is then measured through continued titration to a lower endpoint, capturing the combined concentration of bicarbonate, carbonate, and hydroxide alkalinity present in the sample.
Many facilities follow standard methods published by organizations such as the American Public Health Association to improve consistency and accuracy during alkalinity measurement procedures. Results are commonly reported in mg l as calcium carbonate equivalents.
Digital and Automated Testing Methods
Modern laboratory and industrial operations often use digital instruments to improve measurement consistency and reduce manual error. A ph meter may be integrated into the determination process to identify precise endpoint values during titration procedures.
Automated titration systems are increasingly used in laboratory environments where high sample volumes and repeatable accuracy are required. These systems can improve efficiency by automatically dispensing reagents, recording measurement data, and calculating alkalinity values with minimal operator adjustment.
Because automated systems reduce variability associated with manual measuring, they are often preferred in facilities requiring detailed laboratory reporting and process documentation.
Field Test Kits and Test Strips
Portable field applications often rely on compact test kits and test strips for rapid alkalinity testing outside the laboratory. These tools allow operators to quickly evaluate a water sample and identify changes in alkalinity conditions before larger operational issues develop.
Although field test methods may offer a smaller test range and lower sensitivity compared to laboratory methods, they remain valuable for routine industrial monitoring, preliminary determining procedures, and on-site troubleshooting.
| Testing Method | Common Use | Advantages | Limitations |
|---|---|---|---|
| Manual titration | Routine industrial testing | Accurate, cost-effective | Requires trained handling |
| Automated titration | Laboratory analysis | High accuracy, repeatability | Higher equipment cost |
| Test strips | Field screening | Fast and portable | Lower precision |
Why Accurate Alkalinity Testing Matters
Accurate alkalinity testing is crucial for maintaining stable water chemistry and protecting industrial systems from operational problems. Because alkalinity directly affects buffering capacity and chemical stability, even small changes in alkalinity values can influence treatment performance, equipment reliability, and overall system efficiency.
When alkalinity levels are not properly controlled, industrial systems may experience several issues that negatively affect performance and operating costs.
Problems Associated With Improper Alkalinity Levels
- Scaling caused by calcium carbonate deposits, which can reduce heat transfer efficiency and restrict system flow
- Mineral build up inside industrial equipment, increasing maintenance requirements and shortening equipment life
- Corrosion caused by unstable ph conditions and reduced buffering protection
- Reduced effectiveness of coagulation processes in wastewater treatment applications
- Process instability in biological wastewater treatment systems where bacteria depend on a stable ph environment
- Reduced efficiency during anaerobic digestion processes due to changing alkalinity conditions
- Increased vulnerability to acidic pollutants and external contaminants such as acid rain
High alkalinity water can also create operational challenges if left unmanaged. Excessive alkalinity may contribute to scaling, interfere with treatment chemistry, and increase chemical consumption across industrial systems.
Best Practices for Reliable Alkalinity Measurement
Consistent alkalinity measurement depends on proper testing procedures, equipment maintenance, and sample handling practices. Following established standard methods helps improve accuracy and supports more reliable treatment decisions across industrial and laboratory environments.
Best Practices for Consistent Testing
- Collect representative water samples: Always obtain a clean sample from a consistent location within the system. Contaminated or poorly collected samples can affect the sample’s alkalinity and reduce testing reliability.
- Calibrate testing equipment regularly: A properly calibrated ph meter is essential for accurate endpoint detection during alkalinity determination procedures. Regular calibration also improves measurement consistency across multiple tests.
- Use fresh reagents and clean equipment: Expired reagents or contaminated testing containers can interfere with color change visibility and reduce sensitivity during titration procedures.
- Follow the correct test range and procedure: Different systems may require different testing approaches depending on alkalinity concentration and operating conditions. Following the appropriate method helps improve accuracy when operators measure alkalinity in industrial applications.
- Document and trend alkalinity values over time: Recording results allows operators to identify gradual system changes before they affect efficiency, stability, or equipment performance. Trend analysis also supports better determining and long-term treatment optimization.
How ETI Supports Water Treatment Professionals

Reliable alkalinity testing depends not only on accurate measurement methods, but also on selecting the right treatment approach for each system condition. Because alkalinity directly affects corrosion control, scaling prevention, microbial performance, and overall process stability, many water treatment professionals rely on experienced technical partners to help optimize treatment programs across industrial and wastewater applications.
ETI supports water treatment professionals with customized chemical programs and technical guidance designed to improve system performance and maintain stable water chemistry conditions. Their capabilities support a wide range of applications, including cooling systems, boilers, process water, and wastewater treatment operations where alkalinity control plays a crucial role in protecting industrial equipment and improving operational efficiency.
ETI supports customers through:
- Custom alkalinity boosters designed to maintain buffering capacity and stable ph conditions
- Cooling and boiler treatment programs that help control scaling, corrosion, and mineral build up
- Advanced dispersant technologies that improve suspended solids control and reduce fouling
- Wastewater treatment solutions that support coagulation, flocculation, and biological wastewater treatment performance
- Laboratory analysis, water testing support, and field troubleshooting assistance
- Custom chemical blending for application-specific water chemistry challenges
- Technical guidance for optimizing treatment performance across varying alkalinity conditions
To learn more about ETI’s water treatment services, contact ETI Water to discuss your system requirements and water treatment goals.
Frequently Asked Questions (FAQs)
What is the difference between alkalinity and pH?
Although pH measures how acidic or alkaline water is at a specific moment, alkalinity measures the water’s ability to resist changes in pH. A stable alkalinity level helps maintain chemical balance and protects industrial systems from sudden acid-related fluctuations.
Why is phenolphthalein alkalinity important?
Phenolphthalein alkalinity helps determine the presence of hydroxide and carbonate compounds in a water sample. This information is useful when evaluating scaling potential, treatment efficiency, and the proportion of alkaline compounds present in industrial systems.
Can alkalinity affect biological wastewater treatment?
Yes, alkalinity is vital in biological wastewater treatment because bacteria involved in anaerobic digestion and contaminant breakdown require stable operating conditions. Low alkalinity can reduce process stability and negatively affect treatment performance.
How often should alkalinity testing be performed?
Testing frequency depends on the application, system size, and operating conditions. Many industrial and wastewater treatment facilities perform routine alkalinity testing daily or weekly to maintain accuracy, stability, and consistent treatment performance.
Can environmental conditions influence alkalinity levels?
Yes, environmental factors such as acid rain, carbon dioxide exposure, and acidic pollutants can affect alkalinity conditions in water systems. Monitoring alkalinity helps protect aquatic life, treatment processes, and industrial equipment from changing water chemistry conditions.