In industrial water systems, disinfection and biological control are rarely “set and forget.” Process changes, variable loads, and shifts in water quality can all influence how oxidizing chemicals behave once they enter the system. This is where Oxidation Potential becomes a practical concept for operators and engineers. Often measured as oxidation reduction potential (also called ORP), oxidation potential reflects the overall tendency of an aqueous solution to promote redox reactions, meaning chemical reactions that involve electron transfer between chemical species.
Unlike a single residual test that targets one disinfectant, ORP provides a broader signal of oxidizing or reducing conditions in the water. It is influenced by many variables, including dissolved oxygen, pH, temperature, and the presence of dissolved species such as organic matter, sulfides, or metals. In disinfection control, an ORP trend can help confirm whether oxidizing chemistry is being delivered effectively and whether the system’s demand is changing.
However, oxidation potential can also be misunderstood. ORP is not an absolute measure of disinfectant residual, and it should not be treated as a universal setpoint that applies across all facilities. Instead, it is best used as a supporting measurement that complements residual testing, microbial monitoring, and operational context.
This article explains the fundamentals of oxidation potential, how ORP is measured, why readings shift in real water treatment environments, and how industrial facilities can use ORP data to improve disinfection control with fewer surprises and better chemistry-driven decisions.
What Oxidation Potential Means in Water Systems
In water treatment, oxidation potential describes the tendency of a water sample to drive oxidation reactions or reduction reactions. In simple terms, it answers a practical question: is the water in an overall state that promotes oxidation, or does it favor reduction? This matters because many treatment and disinfection processes depend on how different chemical species behave under oxidizing or reducing conditions.
At the molecular level, the difference between oxidation and reduction is based on electron transfer. During oxidation, a chemical species will lose electrons. During reduction, a chemical species will acquire electrons. This paired relationship is why these processes are called redox reactions, the oxidation step and the reduction step occur together. One substance acts as an electron donor, and another acts as an electron acceptor.
- A substance that donate electrons is a reducing agent because it causes another species to be reduced.
- A substance that accepts electrons is an oxidizing agent because it drives oxidation in another species.
In most industrial water environments, ORP represents the combined effect of many oxidizing and reducing influences. For example, dissolved oxygen often contributes to more oxidizing conditions, while certain reduced compounds, such as sulfides, tend to create reducing conditions. The final ORP signal is shaped by the mix of organic and inorganic compounds, and not just one disinfectant or one contaminant.
This is why oxidation potential is best understood as a “whole-water” indicator. It reflects the balance of oxidation reduction behavior occurring among chemical compounds in solution, including:
- inorganic compounds (such as iron species and other ions)
- organic matter and other carbon-based compounds
- disinfectants and oxidizers that participate in oxidation reduction reactions
- reduced contaminants that consume oxidizers
In practice, ORP is not simply a measure of “how much oxidizer is present.” It is a measurement of how strongly the water environment favors oxidation or reduction overall. Because multiple reactions occur simultaneously, ORP can shift rapidly when system demand changes, when water chemistry changes, or when other substances enter the water.
The Electrochemical Foundation: Half Reactions and Electrodes
To understand how ORP works in the field, it helps to see it as an electrochemical measurement rather than a direct chemical test. ORP instruments do not “count” oxidizer concentration. Instead, they detect an electrical potential difference created by oxidation reduction activity in the water. That potential is generated by two electrodes working together inside the sensor.
ORP is built from two electrodes
An ORP probe uses two electrodes:
- A measuring electrode, typically a platinum electrode, which interacts with the surrounding water and responds to redox activity.
- A reference electrode, which maintains a stable, known potential so the instrument has a baseline for comparison.
When these electrodes are placed in an aqueous solution, they form a small electrochemical cell. The probe output is the voltage difference between the measuring electrode and the reference electrode. This is why ORP is reported in millivolts, the sensor is literally measuring a voltage.
Half reactions: why redox produces voltage
Redox chemistry can be represented using a half reaction, which shows either oxidation or reduction occurring separately. For example:
- In oxidation reactions, a species will lose electrons.
- In reduction reactions, a species will acquire electrons.
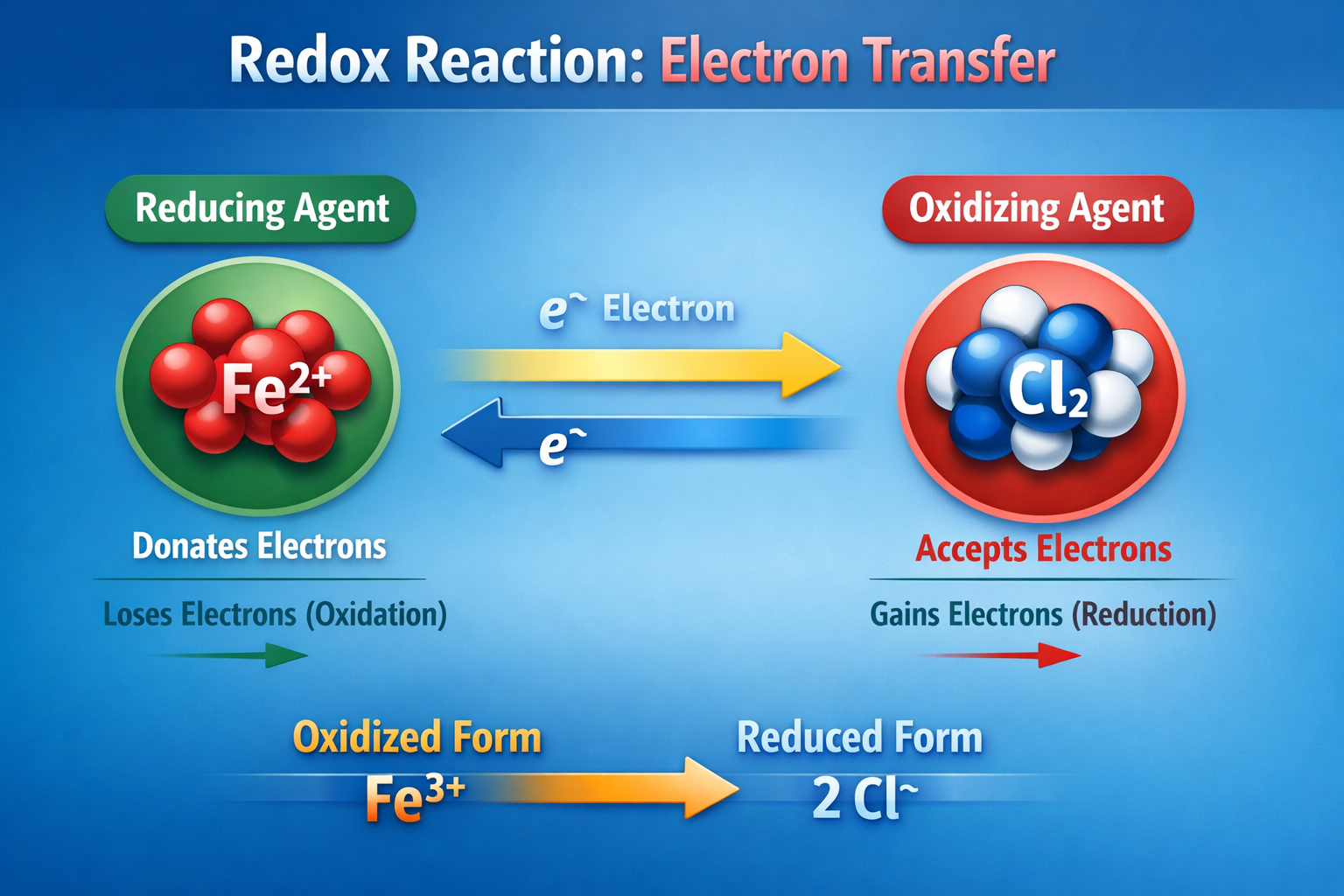
When both occur in the same water sample, electrons are exchanged between chemical species. Even though these are not “free” electrons moving around like in a wire, the surface of the measuring electrode responds to the tendency of dissolved oxidants and reductants to exchange electrons. The resulting signal is the electrical expression of the water’s overall redox environment.
This is also why ORP is sometimes described as a measure of the water’s tendency for oxidation reduction to occur. If the solution contains stronger oxidants, the ORP will generally move in a more positive direction. If the solution contains stronger reducing species, the ORP shifts lower.
The half cell concept in practical terms
The probe functions as a half cell because the measuring electrode surface acts as a site where redox interactions are reflected as electrical potential. The reference electrode completes the measurement by providing a stable comparison point. The ORP meter then converts that voltage signal into a readable value, which is measured and displayed as the ORP reading.
Because this is an electrochemical measurement, ORP responds to a broad range of chemical reactions occurring in the water, not one specific compound. It can also be influenced by ions, coating of the electrode surface, and changes in solution properties.
ORP Reference Standards and the Standard Hydrogen Electrode
Because ORP is an electrical measurement, the value depends on the reference scale used. In electrochemistry, reduction potentials are defined relative to a universal baseline called the standard hydrogen electrode (SHE). Under standard conditions, the SHE is assigned a potential of zero volts. This does not mean “no reaction.” It simply means the hydrogen reference half cell is the agreed starting point for comparing other redox couples.
In a laboratory setting, chemists use SHE to define standard reduction potential values, often written as E° (or e o) in a published table of redox pairs. These values indicate how strongly a given species tends to undergo standard reduction. A more positive E° suggests a stronger tendency to accept electrons and be reduced, while a more negative value indicates a weaker tendency to be reduced. In everyday terms, a species with a negative reduction potential is more likely to act as a reducing species under comparable conditions.
The opposite tendency can be described through standard oxidation potential, sometimes expressed as e ox for an oxidation half reaction. Whether using standard reduction potential or standard oxidation potential, the goal is the same, to compare redox tendencies on a consistent scale.
What ORP Values Tell You, and What They Do Not
ORP readings are often treated like a single “score” for disinfection, but in practice, ORP values are best interpreted as a system-level indicator of overall redox conditions. ORP reflects the combined behavior of many oxidizing and reducing influences in the water, including disinfectants, reduced contaminants, and naturally occurring dissolved species.
What ORP can tell you
In industrial water treatment, ORP is most useful when it is used to understand trends and shifts. It can help operators:
- Track whether oxidizing chemistry is being delivered consistently
- Recognize when demand changes, for example, sudden increases in organic loading
- Identify the onset of reducing conditions, which may correlate with low oxygen or the presence of strong reducing compounds
- Support process troubleshooting when biological control becomes unstable
Because ORP is related to reduction potential ORP, it can serve as an early signal when a water body or system transitions from more oxidizing to more reducing behavior. That transition often indicates changes in water quality, process upsets, or changes in contamination load.
What ORP cannot tell you
ORP is not a replacement for residual testing, microbiological monitoring, or targeted chemical analysis. A common misunderstanding is assuming that a higher ORP means a higher disinfectant residual. ORP is affected by other substances, including organic matter, certain metals, and reduced inorganic species. Two systems can show similar ORP while having very different disinfectant residuals, and the same system can show different ORP readings at the same residual depending on pH and temperature.
In short, ORP is valuable because it integrates many chemical influences into one signal. Its limitation is that it does not isolate individual contributors. The best ORP programs treat the ORP number as an operational trend tool, supported by chemistry and context.
Oxidation Potential in Disinfection Control (Industrial Relevance)
Disinfection and biological control depend on creating conditions that limit the survival and growth of microorganisms. In many industrial systems, oxidizing chemistry is used to disrupt cell structures, damage enzymes, and interfere with metabolic processes. While the specific mechanisms depend on the disinfectant, most programs rely on oxidation reduction behavior, meaning the disinfectant participates in redox chemistry that affects other organisms in the water.
This is where Oxidation Potential becomes a useful operational indicator. Because ORP responds to the balance between oxidizing and reducing chemical activity, it can help confirm whether the system is maintaining sufficiently oxidizing conditions to support disinfection goals. In practice, facilities often use ORP trends to evaluate whether an oxidizer is:
- Reaching the target location in the system
- Being consumed too quickly due to chemical demand
- Maintaining a consistent control response across load changes
Why ORP is not a universal setpoint
Even though ORP is linked to disinfection performance, it is not transferable as a single “magic number” from one site to another. ORP depends on the overall chemistry of the water, including organic and inorganic compounds, pH, dissolved oxygen, and the presence of reducing agents. A water body with higher organic load may show lower ORP even when disinfection is effective, while a cleaner system may show higher ORP at the same disinfectant dosage.
This variability is why ORP should be treated as a facility-specific tool. The objective is to establish a normal operating band based on real operating conditions, then watch for deviations.
ORP and oxygen-driven chemistry
Oxygen plays a central role in redox conditions. Higher oxygen levels generally support more oxidizing conditions, while low oxygen environments often shift toward reducing chemistry. In some systems, these shifts occur due to stagnant zones, high biological activity, or the introduction of reduced contaminants.
A practical example
Consider two cooling systems running the same oxidizer program. If one system receives periodic organic contamination, the oxidizer demand will increase. ORP will often fall, not because the oxidizer feed pump failed, but because the oxidizer is being consumed in competing chemical reactions. In this way, ORP becomes a real-time indicator of changing system demand.
Interferences and Demand: Why ORP Changes in Real Facilities
In real industrial systems, ORP rarely stays steady for long. Even when chemical feed rates remain constant, oxidation reduction conditions can shift because ORP reflects a balance of competing reactions. Any change in the types or concentrations of dissolved species can alter the measured signal, sometimes quickly. This is why ORP is often best used as a trend indicator rather than a single-number target.
The role of oxidizer demand
When oxidizing chemistry is added to a system, it reacts not only with microorganisms, but also with many organic and inorganic compounds. These competing reactions can consume oxidizer and lower the ORP reading, even if the oxidizer is still being fed correctly. Common demand drivers include:
- organic matter, such as process carryover, oils, and biodegradable organics
- reduced sulfur compounds, including hydrogen sulfide
- reduced metals and heavy metals, including certain forms of iron
- reduced nitrogen species, depending on the water source and process conditions
- chemical compounds that act as electron donors in redox chemistry
These materials function as reducing influences, and many act as a reducing agent because they tend to donate electrons in a redox environment. When these reducing compounds enter the system, ORP shifts downward as the solution trends toward reducing conditions.
Hydrogen sulfide and strong reducing behavior
Hydrogen sulfide is a notable example because it is a strong reducing compound in water. Even at low concentrations, it can rapidly consume oxidizers and drive ORP lower. In systems with anaerobic zones or high biological activity, sulfide formation can occur and contribute to unstable ORP trends.
Metals, scaling, and catalytic behavior
Metals such as iron can affect ORP in two ways. First, iron can participate directly in redox reactions as it moves between oxidized and reduced states. Second, iron deposits and other inorganic solids can coat sensors or create microenvironments that alter the local redox behavior. Some metal surfaces can also accelerate oxidation reactions, changing how quickly oxidizers are consumed.
Measuring ORP in the Field: Best Practices
Reliable ORP data depends less on the meter itself and more on how the sensor is installed, maintained, and interpreted. Since ORP is an electrochemical signal, orp measurement quality can be affected by fouling, flow conditions, and changes in solution chemistry. Facilities that treat ORP as a controlled instrument, rather than a “drop-in” probe, tend to get the most useful trends.
How ORP probes measure the signal
To measure ORP, a probe compares the electrical potential at a measuring electrode (often a platinum electrode) against a stable reference electrode. The meter reports the potential difference between these two electrodes as a millivolt value. The reading reflects the net behavior of dissolved oxidants and reductants interacting with the electrode surface in the aqueous solution.
Because ORP responds to many dissolved species, probe placement is critical. Installing the sensor in a well-mixed location helps ensure the reading represents the bulk solution, rather than a stagnant zone or a chemical injection point.
Practical installation and placement guidance
For consistent results:
- Place ORP sensors where the water is fully mixed and flowing
- Avoid dead legs, low-flow side streams, or locations with air entrainment
- Avoid mounting directly downstream of chemical injection where localized chemistry may skew readings
- Ensure temperature conditions are stable, because temperature affects reaction rates and electrode response
Probe care and verification (where most ORP issues occur)
ORP sensors are sensitive to surface condition. A coated or scaled electrode can produce sluggish or inaccurate readings. Regular cleaning and verification are essential, especially in waters with iron, biofilm risk, or high solids.
Recommended practices include:
- Clean the measuring electrode surface according to manufacturer guidance
- Inspect the reference junction for clogging or contamination
- Use a standard ORP check solution to verify that the probe reads within an acceptable tolerance
- Track drift over time, not just “pass or fail” checks
Since ORP is influenced by concentration of dissolved species, changes in the process can also change readings even when the instrument is functioning correctly. Pair ORP trends with key supporting signals such as a pH sensor and conductivity to interpret changes in context.
If your ORP reading is unstable, check these first
- Sensor is located in a low-flow area or poorly mixed zone
- Reference junction is fouled or depleted
- Electrode surface is coated (iron, scale, biofilm)
- Temperature swings or inconsistent flow patterns
- Strong reducing inputs entering the system (organics, sulfides)
- Calibration or verification check solution is expired
The goal is not to chase a perfect number, but to build reliable ORP trends that can be used alongside other measurements to guide chemical control decisions.
ETI’s Role: Chemistry-Driven Oxidation Control Support
In industrial water treatment, controlling oxidation and reduction conditions is rarely about a single chemical. It is about managing the overall balance of reactive chemical species in the system so performance stays stable over time. ETI supports this work by helping facilities and water treatment partners translate ORP trends into practical chemical and operational decisions, including program optimization, troubleshooting, and training.
One area where redox management directly impacts equipment reliability is boiler systems. Dissolved oxygen in boiler feedwater promotes oxidation reactions at metal surfaces, which can lead to pitting, scaling, reduced heat transfer, and premature failure. ETI’s oxygen scavenger programs are designed to remove dissolved oxygen from feedwater, reduce corrosion risk, and support long-term system performance. Depending on operating conditions, ETI can provide sulfite-based scavengers for low to medium pressure boilers, volatile DEHA programs for feedwater and condensate protection, erythorbate-based options for low-sulfate or food-grade needs, and hydrazine alternatives for high-pressure applications.
Alongside product support, ETI emphasizes implementation fundamentals such as correct feed location, dosing verification, and ongoing monitoring so chemical treatment aligns with system demand and water chemistry.
Contact ETI to discuss ORP interpretation, disinfection control strategies, or boiler oxygen scavenger programs tailored to your facility.
Frequently Asked Questions (FAQs)
What is an oxidation potential?
Oxidation potential describes how strongly an aqueous solution favors oxidation, meaning conditions where certain chemical species tend to lose electrons during redox reactions. In industrial water treatment, it reflects the combined influence of organic and inorganic compounds, dissolved metals, and other reactive dissolved species that drive overall oxidation or reduction behavior.
What does ORP tell you?
ORP tells you whether the system is trending toward oxidizing conditions or reducing conditions based on the net balance of redox activity in the water, not a single disinfectant residual. ETI typically uses ORP as a trend tool to identify changing demand from organic matter, hydrogen sulfide, or reduced metals like iron, and to guide troubleshooting alongside other measurements.
How to find oxidation potential?
The practical way to find oxidation potential is to measure ORP using an ORP probe with a platinum electrode (measuring electrode) and a reference electrode, which reports a millivolt potential difference as an ORP measurement. For theoretical comparison, oxidation and standard reduction potential values are referenced to the standard hydrogen electrode at zero volts under standard conditions, but field readings are shaped by temperature, concentration, and other factors.
What do you mean by oxidising potential?
Oxidising potential is another way of describing oxidation potential, and it refers to how strongly water favors oxidation where oxidants accept electrons and other species become oxidized. ETI emphasizes that oxidising potential is influenced by many factors, including dissolved oxygen, pH (via a pH sensor), and interfering compounds, so it should be interpreted as a system signal rather than a universal setpoint.





