In the field of industrial chemistry, the choice of an effective oxidizing agent can determine how well a facility’s water systems perform. Contaminants such as iron, manganese, and organic matter continue to challenge operators in manufacturing, power generation, and other industrial processes. When left unchecked, these impurities can impair efficiency, shorten equipment life, and compromise both safety and compliance. To address these issues, engineers turn to advanced chemical solutions that can oxidize and neutralize contaminants before they cause costly problems.
Among these solutions, sodium permanganate has emerged as a reliable and versatile tool. This deep purple compound belongs to the permanganate family of chemicals and is recognized for its strong reactivity in water treatment. Its ability to oxidize a wide range of contaminants makes it especially valuable where complete and lasting treatment results are essential.
Yet sodium permanganate uses are not the only option available to professionals. Other oxidants such as potassium permanganate, hydrogen peroxide, ozone, and chlorine dioxide are also used across different applications. Each chemical comes with unique strengths, limitations, and safety considerations. This article provides a practical comparison, guiding water treatment engineers through the role of sodium permanganate relative to other oxidants.
Understanding Sodium Permanganate Uses
Sodium permanganate (NaMnO₄) is a highly reactive compound widely recognized in chemistry as a reliable tool for oxidation. It is part of the permanganate family and is often compared with its more familiar counterpart, potassium permanganate. What makes sodium permanganate particularly useful in industrial applications is its unique physical and chemical profile.
This oxidant is supplied as a dark purple liquid, stable in aqueous solutions, and is far more soluble than potassium permanganate. That high solubility allows engineers to prepare concentrated feeds without the challenges of undissolved solids or sediment buildup. In practice, this property simplifies dosing, ensures consistent dispersion, and allows for precise control when treating complex water streams.
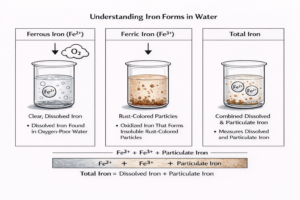
From a chemical standpoint, sodium permanganate is classified as a strong oxidizer. It works by transferring oxygen to contaminants, breaking down both inorganic and organic matter into less harmful byproducts. For instance, it readily oxidizes manganese and iron into insoluble forms that can be filtered out, while also degrading stubborn organic pollutants that might otherwise pass through conventional treatment.
Because it is derived from a salt form of permanganate, sodium permanganate provides stability while maintaining the reactivity that operators need. Its dark coloration, which remains visible until the oxidant is consumed, also offers a built-in monitoring benefit during treatment processes. These combined features make sodium permanganate an important option in the expanding toolbox of modern industrial chemistry.
Sodium Permanganate in Industrial Applications
The versatility of sodium permanganate becomes most evident when looking at its role across diverse industrial applications. Beyond routine water treatment, this oxidant is frequently deployed in specialized sectors where persistent contaminants require a reliable solution.
Groundwater and Soil Remediation
One of the most significant uses is in remediation of contaminated groundwater and soil. The EPA has long recognized sodium permanganate as an effective treatment for chlorinated solvents and other legacy pollutants found at industrial and commercial sites. Its ability to oxidize a wide range of contaminants, including volatile organic compounds, makes it a valuable tool for environmental engineers tasked with restoring impacted areas to safe conditions.
Iron and Manganese Control
Sodium permanganate also plays a role in treating groundwater used within manufacturing facilities. Elevated iron and manganese, common in regions like Pennsylvania and Ohio, can impair operations and reduce water quality. By oxidizing these metals into insoluble forms, sodium permanganate helps provide a complete treatment process that improves both system performance and finished water aesthetics.
Safety and Handling Precautions
While powerful, sodium permanganate must be handled with care. Direct exposure to concentrated solutions can be harmful to human health and damaging to certain materials. Recommended precautions include protective clothing, proper chemical-resistant equipment, and controlled environments for handling. Facilities should also prioritize safe storage conditions, ensuring containers are kept away from incompatible chemicals and maintained in well-ventilated areas.
For engineers and operators, sodium permanganate represents more than a simple chemical—it is a flexible and proven option that, when applied responsibly, supports compliance, safety, and efficiency across a range of industrial water treatment challenges.
Comparing Sodium Permanganate with Other Oxidants
When selecting an oxidizer for industrial water treatment, engineers weigh a range of factors including stability, ease of use, reactivity, and safety. Sodium permanganate is not the only option available, and comparing it with other common oxidants highlights both its advantages and its limits.
Potassium Permanganate
Perhaps the closest relative, potassium permanganate is widely used in drinking water applications to control iron, manganese, and taste issues. Unlike sodium permanganate, it is sold in a crystalline form that must be dissolved before use. While effective, its lower solubility can create challenges in preparing concentrated feeds and maintaining consistent dosing. By contrast, sodium permanganate is a liquid that is easier to manage and delivers better dispersion in treatment systems.
Hydrogen Peroxide
Hydrogen peroxide is another widely used oxidizer, valued for its strong reactivity with organic contaminants. However, it decomposes rapidly, generating heat and oxygen gas that can complicate system operations. This instability makes it less suitable for long-term storage. Sodium permanganate, in comparison, remains stable in solution and can provide predictable performance over time.
Ozone
Ozone is one of the most powerful oxidants available. It is highly effective at breaking down a wide range of contaminants, but it must be generated on-site because it cannot be stored. This requirement adds cost and complexity to treatment systems. While ozone is unmatched in reactivity, sodium permanganate offers a practical alternative for facilities making decisions where simplicity and storage stability are priorities.
Chlorine Dioxide
Chlorine dioxide plays an important role in industrial processes where disinfection and microbial control are needed. However, it poses greater safety risks during handling and requires careful regulatory compliance. In contrast, sodium permanganate offers a safer balance for many industrial chemistry applications, particularly where metals oxidation and contaminant breakdown are the primary goals.
| Oxidant | Form & Solubility | Stability & Storage | Main Applications | Advantages | Limitations |
|---|---|---|---|---|---|
| Sodium Permanganate | Liquid, high solubility, easy dispersion | Stable in storage, predictable dosing | Metals removal (Fe/Mn), remediation, wastewater oxidation | Strong oxidizing agent, built-in monitoring (purple color), safer handling vs. gas oxidants | Higher cost, sodium presence in soil/water |
| Potassium Permanganate | Crystalline solid, lower solubility | Requires on-site dissolving, sediment risk | Drinking water Fe/Mn removal, taste/odor control | Widely available, effective for Fe/Mn | Difficult to make concentrated feeds, dosing inconsistency |
| Hydrogen Peroxide | Liquid, soluble | Decomposes quickly, generates heat & gas | Organics oxidation, disinfection booster | Inexpensive, fast-acting | Short shelf life, handling safety issues |
| Ozone | Gas, generated on-site | Cannot be stored, unstable | Advanced oxidation, strong disinfection | Most powerful oxidizer, highly effective | Expensive equipment, complex operations |
| Chlorine Dioxide | Gas (generated on-site), soluble in water | Requires controlled storage & generation | Microbial control, disinfection | Effective biocide, penetrates biofilms | Handling risks, strict regulatory compliance |
Finding the Right Combination
In practice, no single oxidant fits every application. Many facilities use a combination of oxidizers, selecting the one best matched to the contaminants at hand. Sodium permanganate is often favored where dispersion, stability, and complete oxidation are required, while other oxidants are applied for niche needs. The key is to review site-specific conditions and select the chemistry that aligns with both treatment goals and operational safety.
Also read this blog: Catalyzed vs. Uncatalyzed Oxygen Scavengers: Making the Right Choice
Benefits and Limitations of Sodium Permanganate
Like any product in industrial chemistry, sodium permanganate offers unique advantages but also requires careful consideration of its drawbacks. Engineers evaluating this oxidant should weigh both sides before implementation.
Benefits
- Strong ability to oxidize a wide range of contaminants, from dissolved metals to organic compounds.
- High solubility and liquid form make it easier to handle, dose, and monitor compared with crystalline alternatives.
- Built-in visual indicator: the purple color remains until the oxidant is consumed, helping operators track treatment progress.
- Considered a cleaner option than some oxidants, as its byproducts are generally non-toxic and safe for the environment.
- Long shelf life and stable storage, providing consistency and reliability in ongoing operations.
Limitations
- Presence of sodium can increase salinity in soil or wastewater, which may be a concern for certain remediation or reuse projects.
- Higher cost compared with some other oxidants, which can affect large-scale project budgets.
- As with all strong oxidants, there are safety considerations. Direct contact with concentrated solutions can be harmful to human health, requiring proper protective equipment and handling protocols.
- Must be kept in appropriate storage conditions, away from incompatible chemicals and heat sources.
- Engineers should review data from site-specific conditions before selecting sodium permanganate, ensuring it aligns with treatment goals.
In short, sodium permanganate provides powerful oxidation capacity and operational convenience, but its limitations require thoughtful planning to achieve safe, effective, and sustainable outcomes.
How ETI Supports Industrial Facilities
For facilities weighing the choice of oxidants, from sodium permanganate to alternative chemistries, the key challenge is rarely selecting a single chemical in isolation. In real-world industrial chemistry, treatment success depends on how products are combined, balanced, and optimized for the specific system. That is where ETI delivers unmatched value.
Rather than supplying generic chemicals, ETI specializes in custom blending and program development. Our approach begins with a detailed review of your water chemistry, operating conditions, and regulatory requirements. From there, our technical team designs targeted formulations that incorporate the right oxidants, inhibitors, dispersants, and biocides to create complete solutions. This approach streamlines water treatment by replacing multiple products with a single engineered formulation, reducing both costs and handling complexity.
We also place emphasis on long-term performance. By developing application-specific blends across cooling towers, boilers, closed loops, and wastewater systems, ETI ensures each solution delivers measurable results while protecting your operational rights to efficiency, safety, and compliance.
If you are evaluating oxidants for applications such as metals removal, remediation, or wastewater cleanup, ETI is the partner to consult. Our team combines deep technical information with decades of blending experience, ensuring every program is tailored for success.
Explore ETI’s Custom Blending and Formulations Excellence.
Choosing the Right Oxidant with Confidence
The decision to use sodium permanganate or another oxidizing agent is not one to take lightly. Each chemical has unique properties that make it suitable for specific industrial processes. Sodium permanganate stands out for its stability, solubility, and broad effectiveness in addressing metals, organic matter, and remediation challenges. Other oxidants may serve targeted roles, but sodium permanganate remains one of the most versatile options in modern industrial chemistry.
Ultimately, the most effective programs are those tailored to your facility’s specific needs. That is where ETI’s custom blending expertise ensures the right chemistry, applied in the right way, for lasting results.
Contact ETI today to consult with our technical team and develop a chemical program that fits your exact water treatment challenges.
Frequently Asked Questions (FAQs)
What references exist for sodium permanganate in industrial chemistry?
Standard texts like Ullmann’s Encyclopedia of Industrial Chemistry have shown sodium permanganate’s potential across multiple industries, from soil cleanup to advanced oxidation processes.
Is sodium permanganate essential in every application?
Not always. While it is an essential tool in certain cases, engineers must review data and system needs, as other oxidants may be more suitable depending on site conditions.
Where does the EPA regulate sodium permanganate use?
The EPA, headquartered in Washington, DC, has generally provided guidance on how permanganates are applied in remediation and drinking water treatment programs.
How is sodium permanganate information usually presented?
Technical text and published case studies often face the challenge of making chemistry accessible. Engineers should consult reliable place-based reports and regulatory documents for the most accurate information.
What is the key advantage for water engineers?
The key advantage is its liquid form with high solubility, which makes sodium permanganate easier to dose, safer to handle, and adaptable to a wide range of treatment programs.