Passivation chemicals play a critical role in protecting metal surfaces by enhancing corrosion resistance and extending equipment life. In industrial water systems, where metals are constantly exposed to oxygen, dissolved solids, and varying chemical conditions, the passivation process helps form a stable protective layer that minimizes degradation and preserves system integrity.
At its core, passivation is a chemical treatment designed to modify the outer layer of a metal surface, reducing its chemical reactivity and promoting the formation of a protective oxide layer. This process is especially important for stainless steel equipment, cooling systems, and boiler operations, where maintaining a durable and uniform surface is essential for long-term performance.
What Is the Passivation Process?
The passivation process is a controlled chemical treatment that alters the surface condition of a metal, allowing it to develop a stable and protective barrier against corrosion. Rather than adding a coating, passivation works by enabling natural chemical reactions that form a passive layer directly on the metal itself.
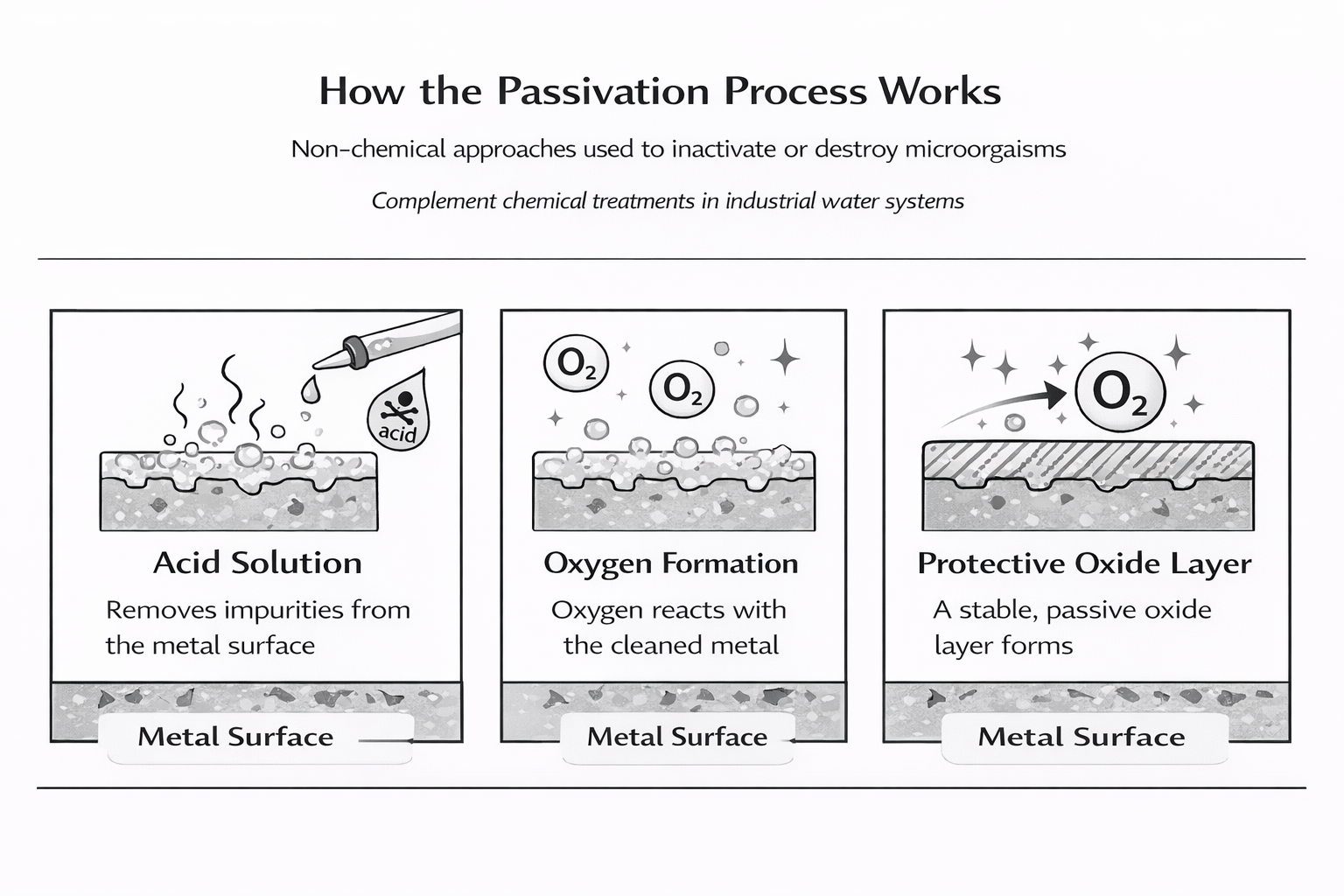
Key Characteristics of a Passive Layer
- The resulting passive layer is chemically non reactive, reducing further corrosion activity
- It consists of an oxide layer that is only a few molecules thick, yet highly effective
- This protective oxide layer adheres tightly to the metal surface, forming a durable barrier against environmental exposure
When properly executed, the passivation process transforms an active surface into a stable metal oxide structure, significantly improving long-term durability and performance.
Passivation of Stainless Steel and Alloys
Stainless steel is widely recognized for its corrosion resistant properties, but its performance depends heavily on proper surface passivation. While stainless steel alloys naturally form a protective chromium oxide layer, this passive oxide layer can be disrupted during fabrication, handling, or exposure to contaminants, making stainless steel passivation a critical step in ensuring long-term reliability.
Why Stainless Steel Requires Passivation
Although stainless steel forms a passive layer on its own, the fabrication process can introduce free iron, foreign material, and surface contamination. These contaminants interfere with the formation of a uniformly corrosion resistant surface and can lead to localized corrosion. Passivating stainless steel helps remove these impurities and restore optimal corrosion resistance.
Role of the Chromium Oxide Layer
The corrosion resistance of stainless steel is driven by the formation of a protective chromium oxide layer on the stainless steel surface. This protective chromium oxide layer acts as a thin yet highly stable surface layer that limits chemical reactivity compared to untreated metal. When properly formed, this passive oxide layer creates a uniform surface that resists oxidation and environmental attack.
Factors That Affect Surface Quality
Several variables influence the effectiveness of stainless steel passivation and the integrity of the resulting passive film:
- Surface contamination from machining, welding, or handling
- Presence of surface grain boundaries where corrosion can initiate
- Residual free iron or iron compounds embedded in the stainless surface
- Variations across different stainless steel forms and stainless steel parts
Proper surface passivation ensures that these factors are addressed, allowing the metal to develop a consistent and durable protective layer that enhances long-term performance.
Nitric Acid vs Citric Acid Passivation
Two of the most widely used approaches to chemical passivation are nitric acid passivation and citric acid passivation. Both methods are designed to remove surface contaminants, promote oxide formation, and create a stable passive film, but they differ in chemistry, handling, and application.
Comparison of Passivation Chemistries
| Factor | Nitric Acid Passivation | Citric Acid Passivation |
|---|---|---|
| Chemistry type | Strong oxidizing acid, often used in a nitric acid bath or concentrated nitric acid solutions | Organic acid used in milder acid bath conditions |
| Mechanism | Nitric acid chemically removes free iron and surface contaminants through aggressive oxidation | Citric acid promotes chemical dissolution of iron compounds and encourages oxygen formation |
| Safety | Requires strict handling, especially in concentrated nitric acid environments | Safer to handle, with fewer hazardous fumes |
| Environmental impact | Higher due to disposal requirements and potential use of sodium dichromate additives | Lower environmental impact, often preferred in modern systems |
| Effectiveness | Highly effective for heavy contamination and industrial applications | Effective for most applications with proper control of acid solution conditions |
| Typical use | Traditional industrial systems, large-scale stainless steel equipment | Food processing, pharmaceutical, and environmentally sensitive applications |
Both methods rely on an acid solution to remove free iron and other surface contaminants that interfere with the formation of a protective passive film. In nitric acid passivation, the oxidizing nature of the acid helps rapidly form a chromium oxide layer, while citric acid passivation works by selectively binding and removing iron, allowing the surface to naturally develop its protective oxide film.
The choice between these methods depends on system requirements, safety considerations, and the desired balance between performance and environmental impact.
Also read: Understanding Condensate Treatment: Oxygen, CO₂, and the Role of Blended Amines
Surface Preparation and Contamination Control
Effective passivation begins well before the chemical treatment itself. Proper surface preparation is essential to ensure that the resulting passive film forms correctly and delivers long-term corrosion resistance. Without removing contaminants and residues, even the best passivation chemicals cannot produce a stable protective layer.
Common Sources of Surface Contamination
- Free iron and exogenous iron transferred during machining or handling
- Foreign material such as oils, greases, and shop debris
- Oxide scale formed during welding or heat treatment
- Residual iron compounds embedded in the metal surface from the fabrication process
These forms of surface contamination interfere with oxide formation and can lead to localized corrosion, even on stainless steel surfaces.
Preparation Steps Before Passivation
- Alkaline cleaning to remove oils, greases, and organic surface contaminants
- Acid solution rinsing to dissolve remaining residues and prepare the surface
- Removal of oxide scale and embedded contaminants to expose a clean underlying material
During preparation, the goal is to remove free iron and any surface contaminants that may disrupt the passivation process. This step ensures that the acid chemically removes unwanted materials without damaging the underlying material.
Also read our guide on: Chelating Agents in Heavy Metal Removal
Passivation in Water Systems and Industrial Applications
In industrial water treatment, passivation chemicals are essential for protecting system materials and ensuring long-term operational reliability. Whether in cooling systems, boilers, or closed loops, the goal is to create a resulting passive layer that shields the underlying material from corrosion and chemical attack.
Cooling Systems
Cooling systems are highly dynamic environments where water chemistry, oxygen levels, and contaminants continuously interact with metal surfaces. Proper passivation helps form a protective layer that reduces chemical reactivity compared to untreated metals. This layer acts to shield material from corrosive conditions while maintaining system efficiency.
Boiler Systems
Boiler systems require tight control of water chemistry to maintain high corrosion resistance. Passivation treatments support the formation of a stable oxide film on internal surfaces, protecting the underlying material from oxygen-related corrosion. When properly managed, this process contributes to consistent performance and extended equipment life.
Closed Loop and Idle Systems
Closed loop systems depend heavily on maintaining a stable passive film, especially since they are often designed to operate with minimal water exchange. Any disruption to the protective layer can expose the underlying material and increase corrosion risk. In idle systems, passivation chemicals help preserve stainless steel equipment and other metals by forming a shield material that limits environmental exposure.
Across all system types, proper passivation ensures that the resulting passive layer provides durable protection, enabling metals to maintain high corrosion resistance even under challenging operating conditions.
Learn more about: Corrosion Inhibitors for Water Treatment Applications
Common Issues and Process Failures
Even with proper materials and chemistry, passivation can fail if the process is not executed correctly. Many corrosion issues in water systems can be traced back to incomplete or ineffective passivation, rather than the performance of ongoing treatment programs.
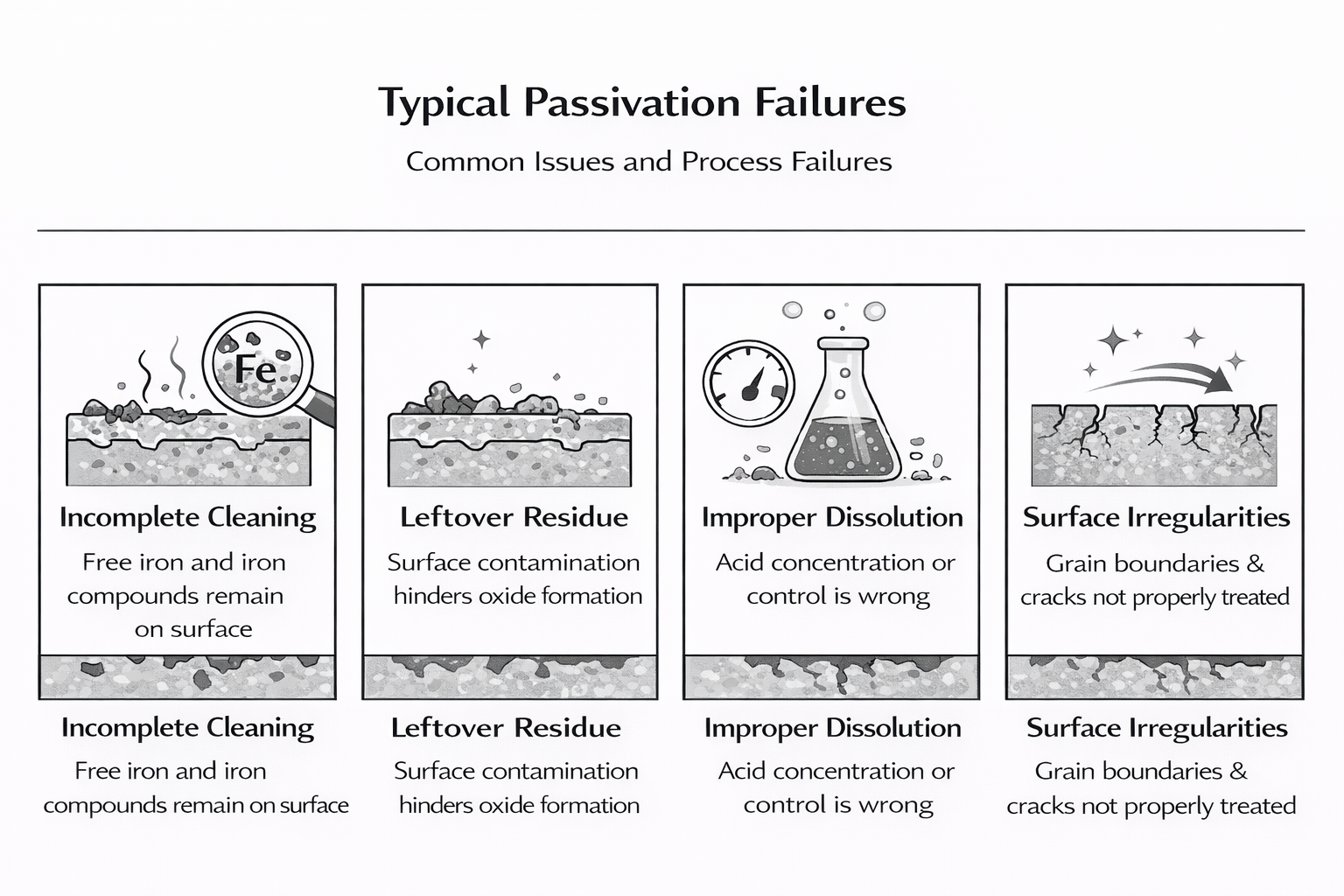
When these issues occur, the passivation process corrects only part of the problem, resulting in an unstable or non-uniform surface. Instead of forming a chemically non reactive surface, the metal remains vulnerable to ongoing corrosion.
Addressing these failures requires careful control of each step, from preparation through final treatment, to ensure that a complete and durable passive layer is achieved.
How ETI Supports Passivation Programs
Passivation is not just about selecting the right chemistry, it requires proper execution, system understanding, and ongoing technical support. This is where ETI plays a critical role for water treatment professionals. As a non-competing partner, ETI provides both passivation chemicals and the technical expertise needed to apply them effectively across a wide range of systems.
How ETI Supports Water Treatment Professionals
- Access to a broad portfolio of treatment chemicals and integrated chemical treatment programs tailored to system requirements
- Technical guidance for system startup, troubleshooting, and optimization of passivation performance
- Support in selecting the right approach for stainless steel equipment, cooling systems, and boiler applications
- Custom formulation capabilities, allowing flexibility in chemical treatment and packaging
- Assistance with safe handling practices such as for acid tank and acid bath applications
ETI’s approach is built around enabling partners to deliver reliable results in the field. With deep technical experience and a commitment to service, ETI helps ensure that passivation programs are executed correctly the first time.
Talk to us to learn more about ETI’s water treatment solutions.
By combining chemistry, technical support, and a partner-first model, ETI equips water treatment professionals with the tools and confidence needed to succeed.
Frequently Asked Questions (FAQs)
What standards apply to stainless steel passivation?
Stainless steel passivation specifications, such as ASTM A967 and AMS 2700, define acceptable methods for achieving a uniformly corrosion resistant surface. These standards ensure that stainless steel parts meet performance requirements for corrosion resistance and cleanliness.
How does passivation remove contamination from metal surfaces?
During chemical passivation, an acid chemically removes free iron, exogenous iron, and other surface contaminants through controlled chemical dissolution. This process exposes the underlying material and allows a protective passive film to form.
What chemicals are used in addition to nitric and citric acid?
In some applications, chemicals such as sodium dichromate or hydrofluoric acid may be used to enhance cleaning or assist in removing oxide scale. These are typically used in specialized acid bath or acid tank processes where additional surface preparation is required.
How thin is the passive film formed during passivation?
The passive film is extremely thin, often only a few molecules thick, yet it provides high corrosion resistance. Despite its size, this thin film creates a stable barrier that protects the metal surface from further chemical reactivity.