Iron is one of the most common elements encountered in industrial water systems. While iron is an essential mineral in biological systems, its presence in cooling water, boiler systems, and process water can create serious operational challenges. Iron can enter water through natural sources, corrosion of piping and equipment, or contamination during industrial processes. Monitoring iron levels helps water treatment professionals detect these issues early and maintain stable system performance.
In industrial applications, excessive iron can contribute to fouling, scaling, and microbiological growth. Particulate iron deposits may accumulate in heat exchangers or piping, reducing efficiency and increasing maintenance costs. Dissolved iron can also promote corrosion reactions that release additional metals into the system, leading to abnormal iron levels that require corrective treatment.
Because of these risks, routine iron testing is a critical part of cooling tower chemistry and treatment programs. By measuring total iron and understanding how iron behaves in water chemistry, water treatment professionals can diagnose system conditions, evaluate treatment performance, and maintain reliable operations across cooling, boiler, and process water systems.
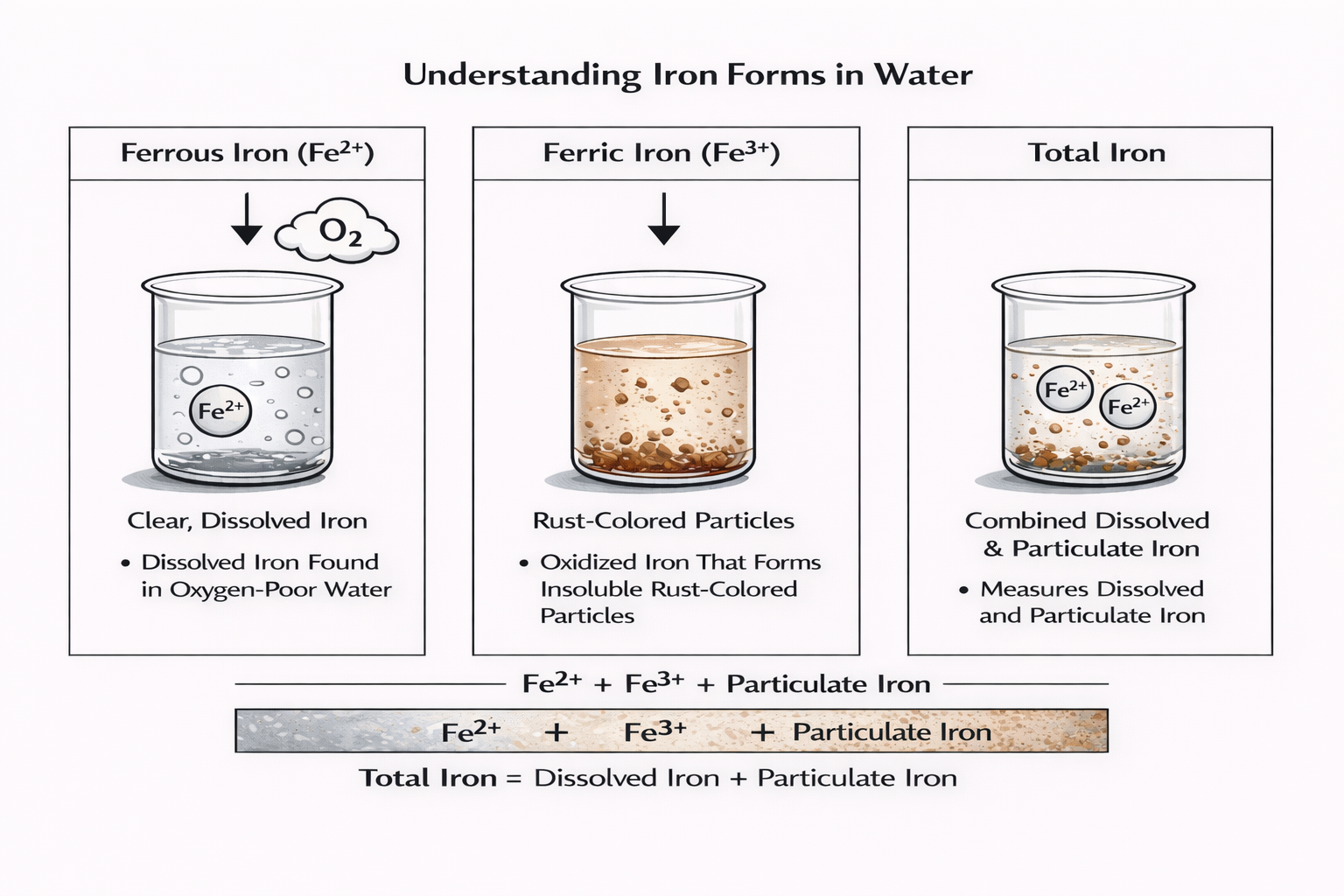
Understanding Iron Forms in Water
Iron in water systems does not exist in just one form. Instead, it can appear in different chemical states depending on oxygen levels, system chemistry, and operating conditions. Understanding these forms is important because iron testing methods often measure specific types of iron, and treatment strategies depend on which form is present.
Ferrous Iron (Fe²⁺)
Ferrous iron is the dissolved form of iron commonly found in oxygen-poor environments such as groundwater or closed systems. It is typically clear in solution and may not be immediately visible in water samples. When exposed to oxygen, ferrous iron quickly oxidizes and can transform into particulate iron deposits.
Ferric Iron (Fe³⁺)
Ferric iron is the oxidized form of iron. It is usually insoluble and often appears as rust-colored particles or sediment in water. Ferric iron can accumulate in filters, piping, or heat transfer equipment, contributing to fouling and operational inefficiencies.
Total Iron
Total iron represents the combined concentration of dissolved and particulate iron present in a sample. Measuring iron total values helps water treatment professionals understand the overall amount of iron in a system, even when iron exists in multiple forms.
| Iron Form | Characteristics | Why It Matters |
|---|---|---|
| Ferrous Iron (Fe²⁺) | Dissolved iron commonly found in low-oxygen environments | Can oxidize quickly and form deposits when exposed to oxygen |
| Ferric Iron (Fe³⁺) | Oxidized, particulate form of iron | Contributes to fouling and sediment accumulation |
| Total Iron | Combined measurement of all iron forms | Provides a complete picture of iron levels in the system |
Identifying whether iron is present as ferrous, ferric, or total iron helps technicians determine whether corrosion, contamination, or process conditions are influencing oxidation-reduction reactions in water treatment. This understanding allows water treatment professionals to select the most appropriate monitoring methods and treatment strategies.
Common Iron Testing Methods Used in Water Treatment
Accurate iron testing is essential for diagnosing corrosion, monitoring treatment performance, and identifying contamination in industrial water systems. Several analytical methods are commonly used in water treatment programs. The choice of method often depends on the required sensitivity, the type of system being monitored, and whether testing is performed in the field or in a laboratory environment.
Phenanthroline Method
The 1,10-phenanthroline method is one of the most widely used approaches in routine iron testing. This colorimetric technique measures ferrous iron by reacting it with a reagent that forms a reddish-orange complex. The color intensity is measured with a photometer or spectrophotometer in clinical chemistry or water testing laboratories.
When ferric iron is present, it is first reduced to ferrous iron before analysis, allowing the test measures to represent total iron in the sample.
Advantages
- Reliable and widely used in routine laboratory tests
- Suitable for portable field instruments
- Provides consistent and repeatable test results
Limitations
- Certain metals or chemicals may interfere with the reaction
- Proper sample collection and reagent preparation are required
Because of its balance of accuracy and practicality, this method is commonly used in industrial water monitoring programs.
Ferrozine Method
The Ferrozine method is another colorimetric technique used to measure dissolved iron. It forms a highly stable purple complex with ferrous iron, which can be measured using a photometer. This method is frequently used in laboratory tests where higher sensitivity is required.
Advantages
- Highly sensitive detection of low iron levels
- Strong selectivity for ferrous iron
- Suitable for detailed analytical work in clinical chemistry environments
The Ferrozine method is often chosen when precise measurements of dissolved iron are necessary.
Atomic Absorption Spectroscopy (AAS)
Atomic Absorption Spectroscopy, commonly called AAS, is a laboratory technique used to measure trace metals in water samples. During analysis, the water sample is atomized in a flame or graphite furnace, and the absorption of light at a specific wavelength indicates the concentration of iron.
AAS is typically performed in specialized analytical laboratories where trained technicians handle sample collection, preparation, and instrument calibration.
Key benefits include:
- High analytical accuracy for measuring iron levels
- Reliable verification of abnormal iron levels
- Strong performance in regulated laboratory tests
Because the method requires specialized equipment, it is generally used for laboratory confirmation rather than routine field monitoring.
Inductively Coupled Plasma (ICP) Analysis
Inductively Coupled Plasma techniques, including ICP-OES and ICP-MS, are among the most advanced methods used for metal analysis. These instruments use extremely high temperatures to excite elements in a sample, allowing analysts to detect very small concentrations of metals.
ICP analysis can measure multiple metals at once, making it valuable when investigating complex water chemistry problems or identifying sources of contamination that may contribute to excess iron or other metal imbalances.
Advantages include:
- Extremely low detection limits
- Simultaneous analysis of multiple metals
- Accurate identification of abnormal iron levels
Because of the cost and complexity of the instrumentation, ICP testing is typically performed in specialized laboratories rather than in routine field operations.
Field Test Kits and Portable Photometers
Many water treatment professionals rely on portable instruments for day-to-day monitoring. These devices use colorimetric reactions similar to laboratory methods but are designed for quick analysis in the field.
Portable systems allow technicians to obtain test results directly at the sampling point, which can help identify changes in iron levels before they affect system performance.
Typical advantages include:
- Fast measurements during routine service visits
- Simple sample collection procedures
- Practical monitoring of iron in your blood levels within water systems, meaning the measurable concentration of iron circulating in the water chemistry
Field testing tools are widely used because they provide rapid feedback that helps technicians respond quickly to developing corrosion or fouling conditions.
Interpreting Iron Test Results in Water Treatment
Conducting iron testing is only the first step in maintaining effective water treatment programs. The real value comes from interpreting test results correctly and understanding what they reveal about system conditions. Changes in measured iron levels often indicate underlying issues such as corrosion, contamination, or filtration performance problems.
Water treatment professionals use routine monitoring to identify trends and respond before equipment performance is affected. Elevated readings may suggest that corrosion is releasing excess iron from piping or equipment, while declining readings may indicate that filtration or treatment adjustments are successfully reducing metal accumulation.
Iron testing results can help identify:
- Corrosion occurring in piping or heat transfer equipment
- Filtration or pretreatment performance issues
- Deposition risks caused by particulate iron
- Changes in system chemistry that lead to abnormal iron levels
Interpreting measurements requires considering system design, operating conditions, and historical monitoring data. Even moderate increases in high iron readings can signal early-stage corrosion or contamination. On the other hand, consistently low iron levels often indicate that treatment programs are effectively controlling corrosion and deposition in troubleshooting dispersant problems in water treatment. This ensures industrial boiler chemistry best practices.
The table below provides a simplified example of how water treatment professionals may interpret common results.
| Iron Concentration | Possible Cause | Recommended Action |
|---|---|---|
| Low Iron | Stable system chemistry and minimal corrosion | Continue routine monitoring |
| Moderate Iron | Early corrosion or contamination entering the system | Investigate potential sources and review treatment program |
| High Iron | Active corrosion, fouling, or filtration failure | Adjust treatment chemistry and inspect system components |
By carefully evaluating iron testing data and comparing results over time, technicians can identify emerging problems early and make informed adjustments to maintain reliable system operation.
Supporting Water Treatment Professionals with Reliable Testing

Reliable iron testing depends not only on the method used, but also on the expertise behind the monitoring program. Water treatment professionals often need technical guidance, reliable products, and troubleshooting support when interpreting test results or responding to changes in iron levels within a system.
Eastern Technologies, Inc. (ETI) supports independent water treatment companies by providing the tools, products, and expertise needed to manage complex water treatment programs. With decades of industry experience, ETI helps partners evaluate system conditions, investigate sources of abnormal iron levels, and select treatment strategies that maintain long-term system reliability.
ETI supports water treatment professionals through:
- Technical guidance for system troubleshooting and monitoring
- Specialized testing tools used in routine water analysis
- Custom water treatment formulations for cooling, boiler, and process water systems
- Reliable manufacturing and distribution capabilities backed by rigorous quality standards
By working as a partner rather than a competitor, ETI enables water treatment professionals to deliver consistent results for their customers while maintaining confidence in their monitoring and treatment programs.
Learn more about how ETI supports water treatment professionals through technical expertise and reliable solutions.
Frequently Asked Questions (FAQs)
Why is iron testing important in industrial water treatment?
Iron testing helps water treatment professionals detect corrosion, contamination, and system fouling before they cause operational problems. Monitoring iron levels allows technicians to identify equipment degradation, evaluate treatment performance, and maintain efficient cooling, boiler, and process water systems.
What is the difference between ferrous iron and ferric iron in water?
Ferrous iron (Fe²⁺) is dissolved iron commonly found in oxygen-poor environments such as groundwater or closed systems. Ferric iron (Fe³⁺) is the oxidized form that forms rust-colored particles and sediment when dissolved iron reacts with oxygen.
What does total iron mean in water testing?
Total iron represents the combined concentration of dissolved iron and particulate iron in a water sample. This measurement helps water treatment professionals determine the overall amount of iron present in a system and evaluate whether corrosion or contamination is occurring.
What are common methods used for iron testing in water systems?
Common iron testing methods include colorimetric tests such as the phenanthroline or Ferrozine method, as well as laboratory techniques like atomic absorption spectroscopy (AAS) and inductively coupled plasma (ICP) analysis. Field test kits and portable photometers are also widely used for routine monitoring.