Across today’s industrial landscape, high purity water is essential to maintaining product integrity, operational efficiency, and regulatory compliance. From biotechnology industries to advanced manufacturing facilities, water that contains even trace levels of contaminants can compromise quality, damage equipment, and interrupt critical applications. In many sectors, the difference between acceptable performance and system failure comes down to how effectively contamination is controlled.
Unlike standard drinking water, which is treated to meet public health standards, high purity water is refined far beyond basic safety requirements. It is engineered to remove dissolved minerals, suspended solids, organic compounds, and microorganisms that could affect sensitive processes. In a world where precision and consistency define success, the ability to deliver and maintain pure water directly impacts customers, facilities, and the broader supply chain.
Understanding how high purity water systems function, and what risks they are designed to prevent, is the first step toward protecting both operational reliability and product quality.
What Is High Purity Water? Definitions and Classifications
In industrial environments, high purity water refers to water that has been treated to remove dissolved ions, organic matter, suspended solids, and microorganisms to levels far below those found in municipal drinking water. The degree of purification depends on the intended application. In biotechnology industries, pharmaceutical manufacturing, electronics production, and specialty chemical manufacture, water quality is often a significant factor in overall process performance.
Water used in these settings is typically prepared through multiple treatment stages and monitored for conductivity, resistivity, total organic carbon, and microbial presence. The goal is not simply to produce clean water, but to produce water with predictable and controlled chemical and biological characteristics.
High purity water systems are designed to achieve this consistency through a combination of filtration, membrane separation, and polishing technologies.
Common Categories of Purified Water
The terminology surrounding pure water can vary by industry, but several classifications are commonly recognized:
| Water Type | Typical Use Case | Key Treatment Approach | Notes |
|---|---|---|---|
| Purified Water | Food processing, laboratory use | Reverse osmosis, filtration, carbon treatment | Removes most dissolved solids and organics |
| Deionized Water | Manufacturing rinse water, process water | Ion exchange resins | Removes calcium, magnesium, sodium, and other ions |
| Sterile Water | Medical and pharmaceutical environments | Filtration and sterilization methods | Free from viable bacteria |
| Ultrapure Water | Semiconductor and critical applications | RO, deionization, UV, degasification, polishing | Extremely low conductivity and TOC |
| Distilled Water | Laboratory and specialty applications | Distillation | Removes many contaminants through phase change |
While drinking water standards focus on safety for human consumption, industrial water standards focus on performance and control. For example, deionized water removes ionic contaminants such as sodium and silica that may interfere with sensitive equipment. Ultrapure water goes even further, targeting trace levels of organic compounds and microorganisms.
These different forms of purified water reflect the specific demands of each application. Selecting the correct grade is a foundational step in protecting process reliability and maintaining consistent product quality.
Core Technologies Behind High Purity Water Systems
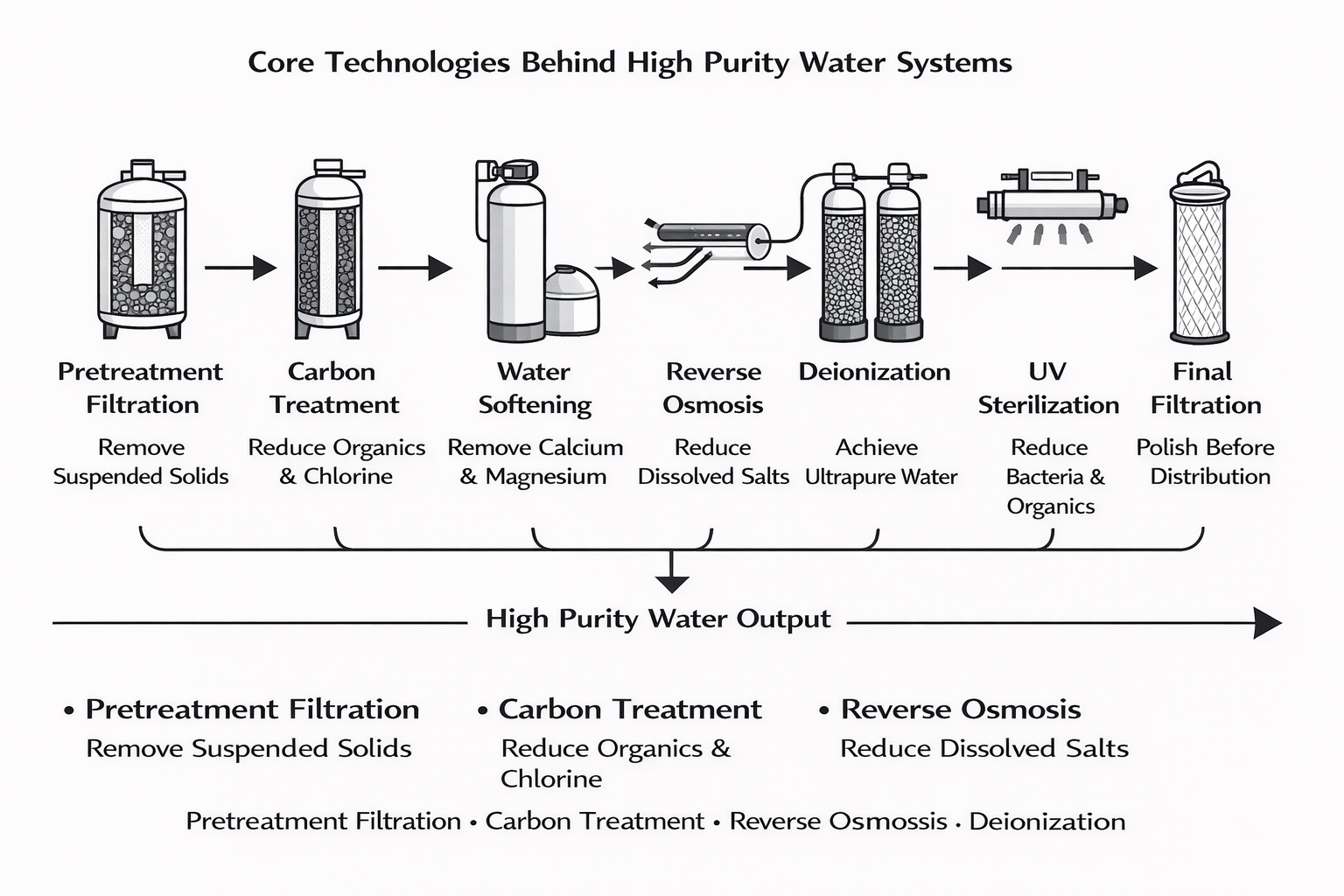
Most high purity water systems rely on a staged treatment process. Each step removes specific contaminants and prepares the water for the next phase. While configurations vary by application and facility requirements, the general process often includes:
- Pretreatment filtration to remove suspended solids and protect downstream equipment
- Carbon treatment to reduce organic contaminants and chlorine
- Water softening to remove calcium and magnesium
- Reverse osmosis to reduce dissolved salts, silica, sodium, and other ionic contaminants
- Polishing with deionization or electrodeionization to achieve higher purity levels
- UV light exposure to reduce bacteria and organic compounds
- Final filtration prior to distribution
This structured approach ensures that no single technology carries the entire burden of purification. Instead, each stage improves overall performance and extends equipment life.
Why Each Stage Matters
Filtration and Pretreatment
Filters protect membranes and downstream components by removing suspended solids, iron, and other particulate matter. Without effective pretreatment, fouling and pressure loss can occur quickly.
Reverse Osmosis
Reverse osmosis uses semi-permeable membranes and applied pressure to separate dissolved contaminants from water. It removes a wide range of impurities, including sodium, silica, and many organic compounds. Proper pressure control is essential for membrane functioning and long-term performance.
Deionization and Polishing
Deionized water is produced by ion exchange resins that remove remaining ionic species. In higher purity applications, polishing systems further reduce conductivity and trace contaminants.
UV Light and Ozone
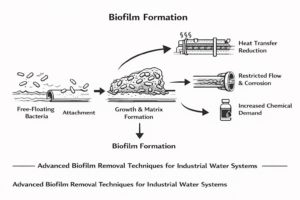
UV light, particularly at specific wavelengths, reduces microorganisms and helps control organic content. Ozone may be used in certain systems to manage bacteria and biofilm risks within storage tanks and distribution loops. Both methods help maintain microbial control without leaving residual chemical contaminants.
Distillation
In select applications, distillation is used to separate water from dissolved solids through evaporation and condensation. This method is energy intensive but effective in removing many contaminants.
Operational Considerations
Beyond the treatment technologies themselves, system performance depends on proper installation, monitoring, and control of temperature, flow rate, and valve operation. Energy efficiency is also an important factor, especially in large facilities where high volumes of water are processed daily.
When properly designed and maintained, high purity water systems provide consistent, reliable water quality for demanding industrial processes.
Common Contaminants and Operational Risks
Contaminant Categories That Matter Most
Even well-designed high purity water systems must continually manage a range of contaminants. These impurities may be present in source water or introduced during storage and distribution. If left unchanged, they can become a major problem for both equipment and product quality.
Key contaminant categories include:
- Dissolved minerals such as calcium and magnesium, which contribute to scale formation
- Silica, which can pass through membranes and deposit on surfaces
- Sodium, often monitored as an indicator of resin exhaustion
- Iron, which can foul membranes and stain equipment
- Suspended solids, which clog filters and increase pressure drop
- Organic compounds, which support microbial growth
- Bacteria and other microorganisms, which can colonize tanks and pipes
Each contaminant behaves differently. Effective control depends on proper system design, routine testing, and consistent sampling at critical points.
Operational Risks in Industrial Facilities
When contamination is not controlled, several operational risks emerge:
- Scaling inside membranes and heat exchange surfaces
- Biofouling from bacteria and microorganisms
- Corrosion of metal pipes and equipment
- Reduced flow rates due to clogged filters
- Increased energy demand caused by higher operating pressure
The following table illustrates how symptoms often connect to root causes:
| Symptom | Likely Cause | First Check |
|---|---|---|
| Rising differential pressure | Fouled filters or scaling | Inspect filtration stage and pressure readings |
| Increased conductivity | Sodium leakage or resin exhaustion | Test deionization performance |
| Visible deposits | Silica or mineral scale | Review pretreatment effectiveness |
| Microbial counts increasing | Biofilm growth in storage loop | Evaluate UV, ozone, and sanitation procedures |
Corrosion within pipes and distribution loops is another concern. Changes in chemistry, oxygen exposure, or stagnant conditions can accelerate material degradation. Over time, this affects overall system functioning and may introduce additional contaminant sources.
Routine testing and documented sampling protocols help identify trends before they escalate. In critical applications, early detection often makes the difference between controlled maintenance and unplanned downtime.
Design, Monitoring, and Expert Operation
Design Choices That Influence Performance
The long-term reliability of high purity water systems depends on thoughtful design and disciplined execution. Several aspects must be evaluated during system development:
- Source water quality and seasonal variation
- Required purity level for the specific process
- Flow rate and peak demand requirements
- Materials of construction for tanks, pipes, and valves
- Temperature control within storage and distribution loops
- Redundancy planning for critical applications
Each decision affects overall functioning and operating cost. For example, improper material selection can increase corrosion risk, while inadequate pretreatment can shorten membrane life. Systems developed with scalability in mind are generally better positioned to adapt as facilities expand or process demands change.
Monitoring and Control Essentials
Maintaining water quality requires continuous monitoring and knowledgeable oversight. Key control methods typically include:
- Conductivity and resistivity measurement
- Routine sampling at defined points
- Microbial testing for bacteria and other microorganisms
- Pressure monitoring across filters and membranes
- Inspection of injection systems used for chemical dosing
Without consistent testing and documented procedures, contamination can go unnoticed until it disrupts production. In controlled environments, maintaining stable temperature and flow conditions also helps avoid contamination and microbial growth.
The Importance of Trained Personnel
Even the most advanced equipment cannot compensate for poor oversight. Expert operation requires trained professionals who understand system dynamics, contaminant behavior, and corrective methods. Knowledgeable operators can interpret data trends, identify irregularities, and implement adjustments before minor deviations escalate.
The cost of neglect is generally far higher than the cost of preventive maintenance. When systems are properly installed, monitored, and maintained, they deliver consistent quality and operational stability. In critical environments where high purity is required, expert management is not optional. It is essential.
Supporting Water Treatment Professionals: ETI’s Role
For companies responsible for designing, maintaining, and optimizing high purity water applications, technical depth and supply reliability make all the difference. Eastern Technologies, Inc. supports independent water treatment professionals and OEM partners through a non-compete, B2B2C model. ETI does not sell direct to end users. Instead, the company equips its customers with the products, expertise, and infrastructure needed to serve their own clients with confidence.
As a 2023 AWT Supplier of the Year and ISO 9001:2015 certified organization, ETI emphasizes quality control, documentation accuracy, and dependable fulfillment. This foundation is especially important in critical applications where contamination risks, compliance requirements, and performance expectations are high.
How ETI Supports High Purity Projects
Water treatment professionals working in industrial facilities often require more than chemical supply. They need a knowledgeable partner capable of supporting complex systems and recent projects systems tailored to specific operating conditions.
ETI provides:
- Technical consultation for system optimization and troubleshooting
- Custom formulation and blending capabilities
- Support for membrane care, cleaning chemistries, and pretreatment control
- Access to the largest team of experienced technical personnel within its distributor network
From New Hampshire to broader national markets, and across Hampshire-based distributors and beyond, ETI focuses on difference serving partners rather than competing with them. The goal is simple: provide dependable service, strengthen customer relationships, and enable long-term growth.
To learn more about ETI’s technical support services, contact us today.
Frequently Asked Questions (FAQs)
How is high purity water different from drinking water?
Drinking water is treated for public health safety, while high purity water is refined to remove dissolved ions, organic compounds, and microorganisms that may affect sensitive industrial processes. The difference serving critical applications lies in tighter control standards and continuous monitoring.
Which industries require ultrapure water?
Ultrapure water is commonly used in biotechnology industries, semiconductor manufacturing, and pharmaceutical production where even trace contamination can impact product quality. These sectors rely on advanced filtration, reverse osmosis, and polishing systems to maintain required purity levels.
What happens if contaminants are left unchanged in a high purity system?
If contaminants such as silica, sodium, or bacteria are left unchanged, they can lead to scaling, corrosion, or biofouling that affects equipment and overall system functioning. Over time, this can become a major problem that increases maintenance costs and disrupts production.
Can high purity water systems be customized for different facilities?
Yes, high purity water systems are generally developed based on source water conditions, required quality levels, and process demands. Proper installation, expert operation, and tailored monitoring methods help ensure the system meets the specific needs of each facility.