In steam and boiler operations, much of the focus is placed on combustion efficiency, boiler feedwater quality, and steam production. However, the health of the condensate return side is just as critical to long-term system efficiency. Without proper condensate treatment, even well-operated systems can experience corrosion, energy losses, and rising maintenance costs.
When steam moves through piping and transfers heat to a process, it eventually cools and condenses back into water. This returning condensate still carries valuable heat and energy, making it an important asset within the overall boiler system. Yet it can also carry dissolved carbon dioxide that contributes to carbon dioxide corrosion through the formation of carbonic acid. If not properly managed, this corrosive environment can damage piping, reduce heat transfer performance, and compromise overall system reliability.
Effective condensate treatment protects steam systems from internal corrosion while preserving efficiency and extending equipment life. It is not simply a chemical adjustment, it is a critical component of long-term operational stability.
How Condensate Forms and Why It Matters
Inside a boiler, water is heated until it becomes saturated steam. This steam carries significant heat content and energy as it travels through piping to support a plant process, provide building heat, or serve direct contact applications. As steam condenses after releasing its heat, it transforms back into hot water, often close to its original condensate temperature. This water formed during phase change is known as condensate.
Although condensate is often compared to distilled water because it has low dissolved solids, it is not chemically neutral. As steam condenses, it can absorb carbon dioxide present in the system. The carbon dioxide content in the vapor phase dissolves into the water, creating conditions that can lead to corrosion if left untreated. This is why condensate treatment is essential to protecting both the boiler and the downstream piping.
When properly managed, returning condensate offers several operational advantages:
- It retains valuable heat content, reducing the need for more fuel to generate more steam
- It lowers the amount of raw make up water required, reducing water required for boiler feedwater preparation
- It helps control dissolved solids in the boiler, improving system efficiency
- It reduces fuel costs and supports overall energy conservation
However, if water losses occur or condensate is not efficiently returned, facilities must introduce more raw make up water. That increases chemical demand, fuel consumption, and operational strain on the entire system.
Carbon Dioxide Corrosion in Condensate Systems
One of the most common and costly threats inside steam condensate systems is carbon dioxide corrosion. When carbon dioxide gas travels with steam and later dissolves into condensate, it reacts with water to form carbonic acid. This weak acid lowers condensate pH and creates an aggressive environment for metal surfaces throughout the return side of the system.
The reaction is straightforward. Carbon dioxide combines with water to form carbonic acid. As condensate moves through return lines and piping, the lowered condensate pH begins attacking the metal surface. Even small reductions in pH can significantly accelerate corrosion rates, particularly in carbon steel components.
Without proper condensate treatment, this process can remain unnoticed until serious damage has already occurred.
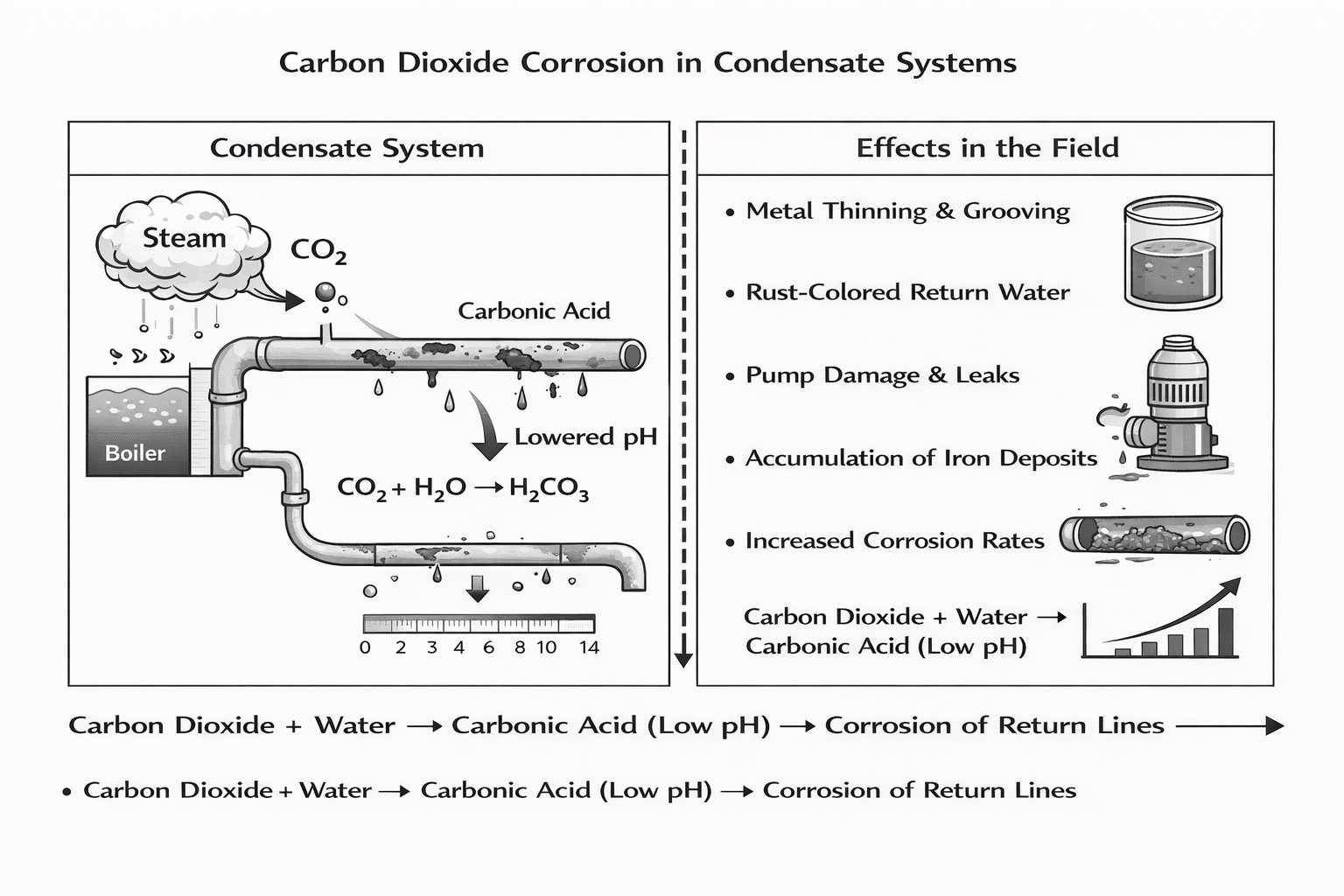
What Carbon Dioxide Corrosion Looks Like in the Field
In many plants, carbon dioxide corrosion develops gradually but produces recognizable patterns:
- General thinning of metal in return lines
- Grooving along the bottom of horizontal piping
- Increased iron levels and corrosion products in returning condensate
- Premature failure of condensate pumps
- Leaks in piping that appear disproportionate to system age
- Accumulation of contaminants and corrosion debris in low-flow areas
Left untreated, corrosion inside steam condensate systems compromises corrosion control efforts in the boiler itself. Iron and other corrosion products are transported back to the boiler feedwater tank, where they can deposit on heat transfer surfaces, reduce efficiency, and contribute to under-deposit corrosion.
Oxygen intrusion can further accelerate damage. Although oxygen scavengers are typically applied on the boiler feedwater side, any oxygen entering through leaks or poor system integrity compounds the corrosive effects of carbonic acid.
You can learn more on this topic in our related article: Understanding Condensate Treatment: Oxygen, CO₂, and the Role of Blended Amines
Treatment Strategies for Effective Corrosion Control
Effective condensate treatment focuses on stabilizing condensate pH, minimizing carbon dioxide corrosion, and protecting metal surfaces throughout the return network. Selecting the right chemical treatment approach depends on system design, operating pressure, condensate temperature, and the most important factor, how steam and condensate move through the facility.
Modern programs typically rely on a combination of treatment chemicals designed to complement the broader boiler feedwater strategy.
Neutralizing Amines
Neutralizing amines are commonly used to control condensate pH. These volatile amines travel with steam, then neutralize carbonic acid when steam condenses. By increasing condensate pH, they reduce corrosion rates in return lines and associated piping.
Neutralizing amines are often preferred when:
- Carbon dioxide content is the primary driver of corrosion
- Systems operate across long distribution distances
- Maintaining target condensate temperature and pH is feasible
- Boiler feedwater alkalinity contributes to carbon dioxide release
Proper feed control is critical. Overfeeding amines can create operational challenges, while underfeeding leaves metal surfaces vulnerable. Monitoring condensate pH and tracking corrosion trends are important factors in maintaining efficient protection.
Filming Amines and Filming Corrosion Inhibitor Programs
Filming amines function differently. Instead of adjusting pH, they form a thin protective barrier on the metal surface. This filming corrosion inhibitor layer reduces direct contact between corrosive condensate and the underlying metal.
Filming programs are especially useful when:
- Corrosion patterns are uneven or localized
- System design includes complex piping layouts
- Oxygen intrusion or other contaminants are present
- Supplemental corrosion control is needed beyond pH adjustment
Because filming amines depend on consistent distribution, careful monitoring is required to avoid potential issues such as overfeed deposition. When properly applied, they support long-term system reliability and efficient performance.
Oxygen Control and the Boiler Feedwater Connection
Although condensate corrosion is largely driven by carbon dioxide, oxygen can intensify damage. Oxygen scavengers applied in boiler feedwater remove dissolved oxygen before steam generation. However, leaks, faulty installation, or poor condensate management can allow oxygen re-entry on the return side.
Maintaining a balanced program requires attention to:
- Dissolved solids levels in boiler feedwater
- Oxygen scavenger performance
- Condensate temperature and flow patterns
- Overall chemical treatment compatibility
An effective condensate treatment strategy must integrate seamlessly with the entire boiler chemical program to maintain corrosion control, protect equipment, and support efficient operation.
Neutralizing vs. Filming Approaches at a Glance
| Approach | Primary Role | Best Fit Conditions | Monitoring Focus | Potential Challenges |
|---|---|---|---|---|
| Neutralizing amines | Raise condensate pH by neutralizing carbonic acid | High carbon dioxide systems with predictable distribution | Condensate pH, amine residual | Overfeed, uneven distribution |
| Filming amines | Create protective barrier on metal surface | Complex systems or localized corrosion patterns | Iron levels, surface protection trends | Deposition if misapplied |
Selecting the correct treatment chemicals requires a clear understanding of system variables and performance goals. When properly designed, condensate treatment enhances corrosion control, protects critical piping, and supports long-term system reliability.
The Role of Condensate Management in System Efficiency
While chemical selection is critical, effective condensate treatment depends equally on strong condensate management practices. Recovering and returning condensate efficiently reduces water losses, conserves energy, and improves overall plant performance.
Every gallon of condensate return carries residual heat. If that heat is not captured and reused, the system must burn more fuel to generate more steam and restore operating temperature. Poor condensate return practices increase fuel costs, raise maintenance costs, and require additional raw make up water to replace water losses.
Beyond energy recovery, proper drainage throughout steam condensate systems helps prevent operational disruptions. Inadequate slope, blocked drain lines, or poorly maintained drain components can allow condensate to pool in piping, increasing corrosion risk and reducing efficiency.
Practical Condensate Management Checkpoints
Facilities can improve system efficiency and reliability by focusing on:
- Ensuring proper drainage throughout piping and return lines
- Inspecting and maintaining each drain line for blockage or restriction
- Monitoring condensate temperature to confirm heat recovery performance
- Reducing water losses to minimize additional water required for boiler feedwater
- Maintaining clean condensate drain pans and associated drain components
- Identifying potential issues in low-flow or idle sections of the system
- Supporting efficient condensate return to the boiler
In some systems, poorly maintained condensate drain pans or stagnant drain pan areas can promote mold growth or algae growth. While these areas are often peripheral to the boiler itself, they still affect overall system cleanliness and maintenance planning. Good housekeeping and drainage practices help prevent mold and support healthier plant environments.
Supporting Water Treatment Professionals: ETI’s Approach
Implementing an effective condensate treatment program requires coordination between boiler chemistry, steam distribution, and condensate return performance. For water treatment professionals managing boiler and steam systems, success depends on having access to reliable treatment chemicals, technical guidance, and responsive manufacturing support.
Eastern Technologies, Inc. supports water treatment professionals with a focused portfolio of boiler treatment solutions designed to address corrosion control, oxygen management, and system protection across low, medium, and high pressure boilers. ETI’s offerings include oxygen scavengers for boiler feedwater, alkalinity builders, dispersants, antiscalants, anti-foam products, and specialty formulations that integrate seamlessly with condensate treatment strategies.
Because condensate corrosion is directly influenced by boiler chemistry, ETI emphasizes program integration. Neutralizing amines, filming amines, and other corrosion inhibitor technologies must align with:
- Boiler feedwater alkalinity and carbon dioxide content
- Oxygen scavenger performance
- System pressure and steam distribution characteristics
- Condensate return rates and temperature profiles
In addition to supplying treatment chemicals, ETI provides technical support to help water treatment professionals evaluate corrosion trends, interpret condensate pH data, and adjust programs based on system design and operating conditions. Custom formulation capabilities allow partners to tailor solutions to specific plant requirements while maintaining system reliability.
To discuss your condensate treatment needs or explore partnership opportunities, contact Eastern Technologies, Inc. and connect with our technical team.
Frequently Asked Questions (FAQs)
Why is condensate pH important?
Condensate pH directly affects corrosion rates on metal surfaces. If pH drops due to carbonic acid formation, corrosion products increase and long-term damage can occur.
What happens if condensate systems are left untreated?
If left untreated, condensate systems can experience serious damage to piping and return lines due to carbon dioxide corrosion and contaminants, increasing maintenance and system downtime.
How do condensate drain pans affect system performance?
Poorly maintained condensate drain pans or a stagnant drain pan can promote mold growth and algae growth. Proper drainage and cleaning help prevent mold and support cleaner system operation.
How does condensate temperature influence corrosion?
Higher condensate temperature can increase corrosion rates if chemical protection is inadequate. Monitoring temperature and treatment levels helps maintain efficient corrosion control.